Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(1):259-270. doi:10.7150/ijbs.53795 This issue Cite
Research Paper
NETO2 promotes esophageal cancer progression by inducing proliferation and metastasis via PI3K/AKT and ERK pathway
1. Endoscopy Center and Endoscopy Research Institute, Zhongshan Hospital, Fudan University, No. 180 FengLin Road, Shanghai 200032, China.
2. Department of Emergency Surgery, First Affiliated Hospital of Nanchang University, Nanchang, China.
3. Endoscopy Center, East Hospital, Tongji University, Shanghai, China.
*These authors contributed equally to this work.
Abstract
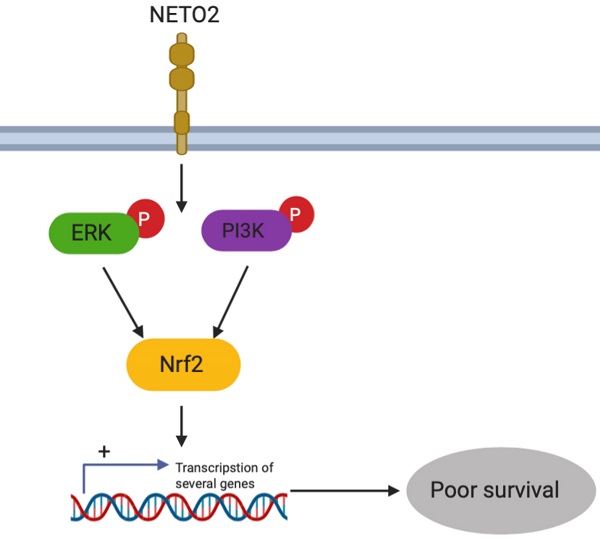
Esophageal squamous cell carcinoma (ESCC) causes aggressive and lethal malignancies with extremely poor prognoses, and accounts for about 90% of cases of esophageal cancer. Neuropilin and tolloid-like 2 (NETO2) protein coding genes have been associated with various human cancers. Nevertheless, little information is reported about the phenotypic expression and its clinical significance in ESCC progression. Here, our study found that NETO2 expression in ESCC patients was associated with tumor clinical stage and lymph node metastasis status. Gain-of-function and loss-of-function analyses showed that NETO2 stimulated ESCC cell proliferation while suppressing apoptosis in vitro and enhanced tumor growth in vivo. Moreover, knockdown of NETO2 significantly inhibited migration and invasion in combination with regulation of epithelial-mesenchymal transition (EMT) related markers. Mechanistically, overexpression of NETO2 increased the phosphorylation of ERK, PI3k/AKT, and Nuclear factor erythroid-2-related factor 2(Nrf2), whereas silencing NETO2 decreased the phosphorylation of these targets. Our data suggest that Nrf2 was a critical downstream event responsible for triggering the PI3K/AKT and ERK signaling pathways and plays a crucial role in NETO2-mediated tumorigenesis. Taken together, NETO2 acts as an oncogene and might serve as a novel therapeutic target or prognostic biomarker in ESCC patients.
Keywords: Esophageal squamous cell carcinoma, NETO2, Proliferation, Metastasis, PI3K/AKT and ERK

 Global reach, higher impact
Global reach, higher impact