Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(1):353-367. doi:10.7150/ijbs.51309 This issue Cite
Research Paper
CDH1 is Identified as A Therapeutic Target for Skin Regeneration after Mechanical Loading
a Department of Plastic & Reconstructive Surgery, Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine, 639 Zhizaoju Road, Shanghai 200011, P.R. China.
#These authors contributed equally to this work
Abstract
Rationale: Mechanical stimuli in the microenvironment are considered key regulators of cell function. Clinically, mechanical force (tissue expander) is widely used to regenerate skin for post-burn or trauma repair, implying that mechanical stretching can promote skin cell regeneration and proliferation. However, the underlying mechanism remains unknown.
Methods: Microarray analysis was utilized to detect the hub gene. The expression of Cdh1 as examined in cells and tissues by western blot, q-PCR and immunohistochemistry staining respectively. Biological roles of Cdh1 was revealed by a series of functional in vitro and in vivo studies.
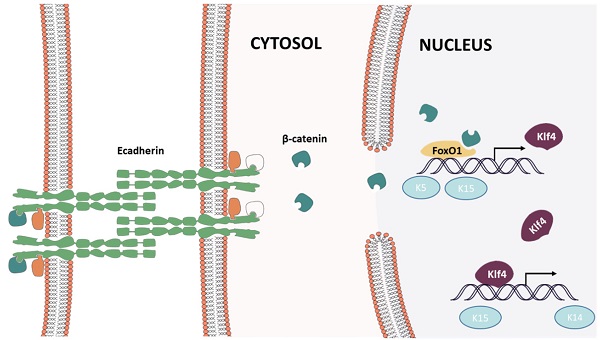
Results: Microarray analysis identified Cdh1 as a hub gene related to skin regeneration during rat cutaneous mechanical loading. In vitro studies suggested that both mechanical loading and Cdh1 interference induced keratinocyte dedifferentiation and enhanced stemness, promoting cell proliferation and prevent apoptosis. Furthermore, the forkhead box O1/Krüppel-like factor 4 (FOXO1/KLF4) pathway was activated and contributed to the keratinocyte dedifferentiation. In vivo studies showed that mechanical loading and Cdh1 interference facilitated epidermal dedifferentiation and promoted dermal collagen deposition, and that Cdh1 overexpression could block such influence.
Conclusions: In this study, we show that E-cadherin (CDH1), a well-known cell-cell adhesion molecule, plays a crucial role in mechanical stretch-induced skin cell regeneration and proliferation. We have shown for the first time the process by which mechanical stress is transmitted to the epidermis and induces a downstream signaling pathway to induce epidermal cells to differentiate. These findings demonstrate that Cdh1-induced keratinocyte dedifferentiation is a crucial event in mechanical stretch-mediated skin regeneration and that Cdh1 may serve as a potential therapeutic target for promoting skin regeneration.
Keywords: skin regeneration, tissue expansion, WGCNA, Mechanical stimuli

 Global reach, higher impact
Global reach, higher impact