Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(2):417-429. doi:10.7150/ijbs.54194 This issue Cite
Research Paper
SOX13/TRIM11/YAP axis promotes the proliferation, migration and chemoresistance of anaplastic thyroid cancer
Department of Thyroid and Breast Surgery, Zhongnan Hospital of Wuhan University, Wuhan, China.
Abstract
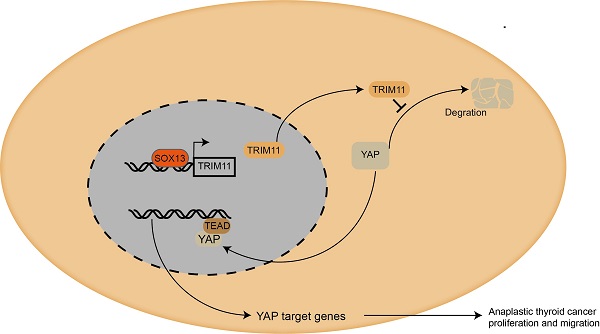
Anaplastic thyroid cancer (ATC) is one of the most aggressive and virulent solid tumors. The ubiquitin proteasome system presents in all eukaryotic cells and is essential for cellular homeostasis. While its underlying role in ATC remains largely unclear. TRIM11 is an E3 ubiquitin ligase and has been reported to act as an oncogene in several human cancers. The present study aims to reveal the oncogenic function of TRIM11 in ATC. Western blot was used to measure the protein expression of TRIM11 and YAP, while the YAP target genes were measured by real-time PCR. CCK8 assay was used to detect cell viability; wound-healing assay and transwell assay were used to measure the migration ability of ATC. The xeno-graft tumor model was used for in vivo study. Immuno-precipitation assay was used to detect the interaction domain between YAP and TRIM11. And the ubiquitin-based Immuno-precipitation assays were used to detect the specific ubiquitination manner happened on YAP. TRIM11 depletion significantly decreases cell proliferation and migration capabilities of ATC cells, and elevates cell sensitivity to chemotherapy, which effect could be further rescued by YAP overexpression. TRIM11 depletion decreases YAP protein level and YAP/TEAD target genes, such as CTGF, ANKRD1 and CYR61 in ATC. Indicating that TRIM11 is a regulator of Hippo signaling pathway. Immuno-precipitation assay shows that the RING domain of TRIM11 is essential for the interaction with WW domain of YAP. Further mechanistic analysis suggests that TRIM11 promotes the mono-ubiquitination of YAP, thus prolongs its protein half. Furthermore, TRIM11 promoter analysis revealed that SOX13 activates TRIM11 transcription by binding to the promoter of TRIM11. In summary, our study describes the oncogenic function of TRIM11 in ATC, which acts as a post-translational modulating factor of Hippo pathway. Targeting TRIM11 may be a potential therapeutic method for ATC treatment.
Keywords: anaplastic thyroid cancer, TRIM11, YAP, stabilization, mono-ubiquitination

 Global reach, higher impact
Global reach, higher impact