Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(3):670-682. doi:10.7150/ijbs.56128 This issue Cite
Research Paper
Basal-like breast cancer with low TGFβ and high TNFα pathway activity is rich in activated memory CD4 T cells and has a good prognosis
1. Bluewater Biotech LLC, New Providence, NJ, USA.
2. Division of Cancer Research and Training, Department of Internal Medicine, Charles Drew University of Medicine and Science, David Geffen UCLA School of Medicine and UCLA Jonsson Comprehensive Cancer Center, Los Angeles, CA 90095, USA.
Abstract
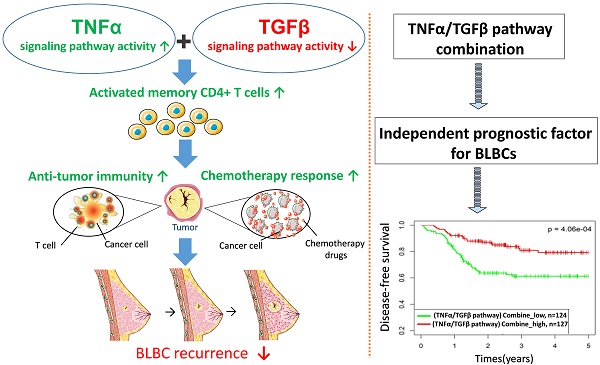
Basal-like breast cancer (BLBC) is a type of high-grade invasive breast cancer with high risk of recurrence, metastases, and poor survival. Immune activation in BLBC is a key factor that influences both cancer progression and therapeutic response, although its molecular mechanisms are not well clarified. In this study, we examined five cancer immunity-related pathways (IFNα, IFNγ, STAT3, TGFβ and TNFα) in four large independent breast cancer cohorts (n = 6,381) and their associations with the prognosis of breast cancer subtypes. Activities of the 5 pathways were calculated based on corresponding pathway signatures and associations between pathways and clinical outcomes were examined by survival analysis. Among the five PAM50-based subtypes, BLBC had the highest IFNα, IFNγ, TNFα pathway activities, and the lowest TGFβ activity. The IFNα, IFNγ, TNFα pathway activities were negatively correlated with BLBC recurrence. In contrast, positive association and no association with BLBC recurrence were observed for TGFβ and STAT3 pathways, respectively. TNFα/TGFβ pathway combination improved the prediction of recurrence and chemotherapy response of BLBCs. Immune cell subset analysis in BLBC showed that M0, M1 and M2 macrophage levels were associated with either TNFα or TGFβ pathways, whereas the level of activated memory CD4 T cells were associated with both pathways. Moreover, this T cell subset was most abundant in BLBCs with low TGFβ and high TNFα pathway activities. These results suggested that cooperation of TNFα and TGFβ signaling may be involved in the regulation of memory T cells and anti-cancer immunity in BLBCs. Our data also demonstrate that TNFα/TGFβ pathway combination may represent a better biomarker for BLBC prognosis and clinical management.
Keywords: Basal-like breast cancer, immunity-related pathways, chemotherapy, prognosis

 Global reach, higher impact
Global reach, higher impact