Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(7):1757-1768. doi:10.7150/ijbs.59001 This issue Cite
Research Paper
Long Noncoding RNA KCNQ1OT1 is a Prognostic Biomarker and mediates CD8+ T cell exhaustion by regulating CD155 Expression in Colorectal Cancer
1. Department of General Surgery, The First Affiliated Hospital of Jinan University, Guangzhou, 510632, China.
2. College of Pharmacy, Jinan University, Guangzhou, 510632, China.
3. Department of Bone and Joint Surgery, The First Affiliated Hospital of Jinan University, Guangzhou, 510630, China.
4. MOE Key Laboratory of Tumor Molecular Biology and Key Laboratory of Functional Protein Research of Guangdong Higher Education Institutes, Institute of Life and Health Engineering, Jinan University, Guangzhou, 510630, China.
#These authors contributed equally to this work.
Abstract
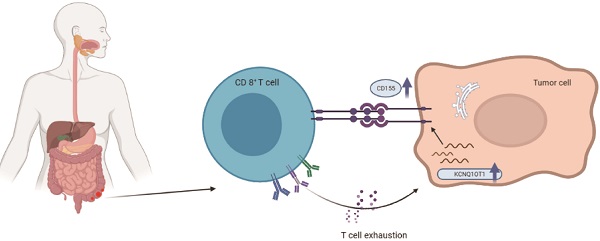
Background: Long noncoding RNA KCNQ1 opposite strand/antisense transcript 1 (lncRNA KCNQ1OT1) is abnormally expressed in various solid tumors. The purpose of this study was to explore the prognostic value and potential functional role of lncRNA KCNQ1OT1 across cancers.
Methods: We performed a meta-analysis of published literature to evaluate the prognostic value of lncRNA KCNQ1OT1 across cancers. Verification, functional analysis, and genomic variation analysis were performed using the GEPIA, TIMER, and LnCeVar databases. According to the immune cell infiltration level, we established a prognostic model of lncRNA KCNQ1OT1 expression using public datasets of TIMER. We used quantitative real-time polymerase chain reaction (RT-qPCR) and western blot to detect the expression levels of lncRNA KCNQ1OT1 and the CD155 protein in colorectal cancer (CRC) tissues and cell lines. Then, a lncRNA KCNQ1OT1-knockdown cell line was cocultured to explore the role of lncRNA KCNQ1OT1 and CD155 in the T cell response by flow cytometric analysis.
Results: Our results showed that the high expression of lncRNA KCNQ1OT1 was significantly related to poor overall survival across cancers, especially CRC. Interestingly, we found that COAD patients with high lncRNA KCNQ1OT1 expression and high CD8+ T cell infiltration levels had a worse prognosis than those with low lncRNA KCNQ1OT1 expression and high CD8+ T cell infiltration levels. Moreover, lncRNA KCNQ1OT1 and CD155 showed significantly higher expression in CRC tissue than in normal tissue, and lncRNA KCNQ1OT1 expression was positively correlated with CD155 expression in CRC. Finally, knockdown of lncRNA KCNQ1OT1 reduced CD155 expression in HCT116 and SW620 cells and enhanced the immune response in coculture with CD8+ T cells.
Conclusions: High lncRNA KCNQ1OT1 expression is significantly correlated with poor prognosis of CRC patients and mediates the CD8+ T cell response in CRC. These findings indicate that lncRNA KCNQ1OT1 is a prognostic biomarker and potential immune therapeutic target for enhancing the CD8+ T cell response in CRC.
Keywords: lncRNA KCNQ1OT1, cancer, prognosis, bioinformatics, tumor-infiltrating, T cell exhaustion

 Global reach, higher impact
Global reach, higher impact