Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(9):2193-2204. doi:10.7150/ijbs.57130 This issue Cite
Research Paper
Dual-specificity Phosphatase 9 protects against Cardiac Hypertrophy by targeting ASK1
1. Department of Cardiovascular Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
2. Department of Anesthesiology, The Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology.
3. Department of Cardiology, The Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
#These authors contributed equally to this work.
Abstract
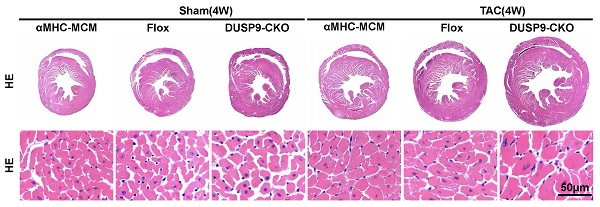
The functions of dual-specificity phosphatase 9 (DUSP9) in hepatic steatosis and metabolic disturbance during nonalcoholic fatty liver disease were discussed in our prior study. However, its roles in the pathophysiology of pressure overload-induced cardiac hypertrophy remain to be illustrated. This study attempted to uncover the potential contributions and underpinning mechanisms of DUSP9 in cardiac hypertrophy. Utilizing the gain-and-loss-of-functional approaches of DUSP9 the cardiac phenotypes arising from the pathological, echocardiographic, and molecular analysis were quantified. The results showed increased levels of DUSP9 in hypertrophic mice heart and angiotensin II treated cardiomyocytes. In accordance with the results of cellular hypertrophy in response to angiotensin II, cardiac hypertrophy exaggeration, fibrosis, and malfunction triggered by pressure overload was evident in the case of cardiac-specific conditional knockout of DUSP9. In contrast, transgenic mice hearts with DUSP9 overexpression portrayed restoration of the hypertrophic phenotypes. Further explorations of molecular mechanisms indicated the direct interaction of DUSP9 with ASK1, which further repressed p38 and JNK signaling pathways. Moreover, blocking ASK1 with ASK1-specific inhibitor compensated the pro-hypertrophic effects induced by DUSP9 deficiency in cardiomyocytes. The main findings of this study suggest the potential of DUSP9 in alleviating cardiac hypertrophy at least partially by repressing ASK1, thereby looks promising as a prospective target against cardiac hypertrophy.

 Global reach, higher impact
Global reach, higher impact