Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(9):2348-2355. doi:10.7150/ijbs.57810 This issue Cite
Research Paper
Capivasertib restricts SARS-CoV-2 cellular entry: a potential clinical application for COVID-19
1. State Key Laboratory of Virology and Modern Virology Research Center, College of Life Sciences, Wuhan University, Wuhan, 430072, China.
2. Sino German Joint Research Center for Agricultural Biology, and State Key Laboratory of Crop Biology, College of Life Sciences, Shandong Agricultural University, Tai'an, 271018, China.
3. Institute of Translational Medicine, Faculty of Health Sciences, University of Macau, Avenida de Universidade, Taipa, Macau SAR; MoE Frontiers Science Center for Precision Oncology, University of Macau, Avenida de Universidade, Taipa, Macau SAR.
4. Hubei Province Key Laboratory of Allergy and Immunology, School of Basic Medical Sciences, Wuhan University, Wuhan 430071, China.
5. Aix-Marseille University, Institute of NeuroPhysiopathology, UMR 7051, 27, Bd Jean Moulin, 13385 Marseille cedex, France.
6. Department of Geriatric Medicine and Memory Clinic, Research Center on Autonomy and Longevity, University Hospital, Angers, France.
7. School of Sciences and Humanities, Biology Department, and Pilot Cluster of Multidisciplinary Comprehensive Materia Medica, Biocluster within Cluster of Life Science and Engineering at C4, Nazarbayev University, Nur-Sultan, 010000, Republic of Kazakhstan.
#Co-first authors.
Abstract
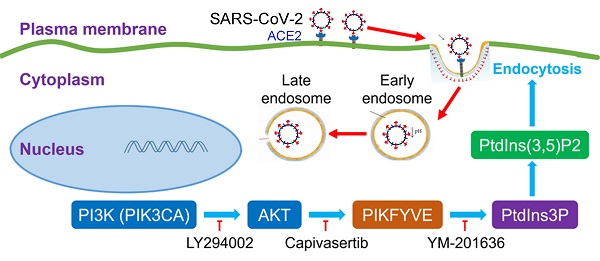
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has led to more than 150 million infections and about 3.1 million deaths up to date. Currently, drugs screened are urgently aiming to block the infection of SARS-CoV-2. Here, we explored the interaction networks of kinase and COVID-19 crosstalk, and identified phosphoinositide 3-kinase (PI3K)/AKT pathway as the most important kinase signal pathway involving COVID-19. Further, we found a PI3K/AKT signal pathway inhibitor capivasertib restricted the entry of SARS-CoV-2 into cells under non-cytotoxic concentrations. Lastly, the signal axis PI3K/AKT/FYVE finger-containing phosphoinositide kinase (PIKfyve)/PtdIns(3,5)P2 was revealed to play a key role during the cellular entry of viruses including SARS-CoV-2, possibly providing potential antiviral targets. Altogether, our study suggests that the PI3K/AKT kinase inhibitor drugs may be a promising anti-SARS-CoV-2 strategy for clinical application, especially for managing cancer patients with COVID-19 in the pandemic era.
Keywords: COVID-19, SARS-CoV-2, AKT inhibitor, capivasertib, antiviral activity

 Global reach, higher impact
Global reach, higher impact