Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(10):2576-2589. doi:10.7150/ijbs.58776 This issue Cite
Research Paper
Aberrant FGFR4 signaling worsens nonalcoholic steatohepatitis in FGF21KO mice
1. Department of Surgery, School of Medicine, University of Louisville, Louisville, KY 40202, USA.
2. Department of Hepatobiliary and Pancreatic Surgery, The First Hospital of Jilin University, Changchun 130021, China.
3. Department of Pathophysiology, Basic Medicine College, China Medical University, Shenyang 110122, China.
4. Department of Tumor Center, The First Hospital of Jilin University, Changchun, Jilin, China.
Abstract
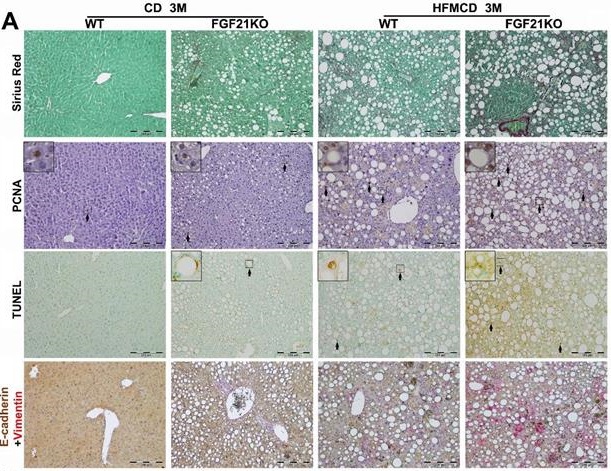
Background: Nonalcoholic steatohepatitis (NASH) is the most severe form of non-alcoholic fatty liver disease (NAFLD) and a potential precursor of hepatocellular carcinoma (HCC). In our previous studies, we found that endocrine fibroblast growth factor 21 (FGF21) played a key role in preventing the development of NASH, however, the FGF15/19 mediated-FGFR4 signaling worsened NASH and even contributed to the NASH-HCC transition. The aim of this study is to determine whether FGF15/FGFR4 signaling could alleviate or aggravate NASH in the FGF21KO mice.
Methods: NASH models were established in FGF21KO mice fed with high fat methionine-choline deficient (HFMCD) diet to investigate FGF15/FGFR4 signaling during early stage NASH and advanced stage NASH. Human hepatocytes, HepG2 and Hep3B cells, were cultured with human enterocytes Caco-2 cells to mimic gut-liver circulation to investigate the potential mechanism of NASH development.
Results: Significant increase of FGF15 production was found in the liver of the NASH-FGF21KO mice, however the increased FGF15 protein was unable to alleviate hepatic lipid accumulation. In contrast, up-regulated FGF15/19/FGFR4 signaling was found in the FGF21KO mice with increased NASH severity, as evident by hepatocyte injury/repair, fibrosis and potential malignant events. In in vitro studies, blockage of FGFR4 by BLU9931 treatment attenuated the lipid accumulation, up-regulated cyclin D1, and epithelial-mesenchymal transition (EMT) in the hepatocytes.
Conclusion: The increased FGF15 in NASH-FGF21KO mice could not substitute for FGF21 to compensate its lipid metabolic benefits thereby to prevent NASH development. Up-regulated FGFR4 signaling in NASH-FGF21KO mice coupled to proliferation and EMT events which were widely accepted to be associated with carcinogenic transformation.
Keywords: Fibroblast growth factor 15/19, Fibroblast growth factor 21, Nonalcoholic steatohepatities, Nonalcoholic fatty liver disease, Bile acid.

 Global reach, higher impact
Global reach, higher impact