Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(13):3672-3688. doi:10.7150/ijbs.60110 This issue Cite
Research Paper
Endophilin A2-mediated alleviation of endoplasmic reticulum stress-induced cardiac injury involves the suppression of ERO1α/IP3R signaling pathway
1. The Fifth Affiliated Hospital, Key Laboratory of Molecular Target & Clinical Pharmacology and the State & NMPA Key Laboratory of Respiratory Disease, School of Pharmaceutical Sciences, Guangzhou Medical University, Guangzhou, 511436, P.R. China.
2. Department of Gastrointestinal Surgery, First Affiliated Hospital of Jinan University, Guangzhou 510630, P.R. China.
3. Guangzhou Institute of Cardiovascular Disease, Guangzhou Key Laboratory of Cardiovascular Disease, and the Second Affiliated Hospital, Guangzhou Medical University, Guangzhou 510260, P.R. China.
#These authors contributed equally to this article.
Abstract
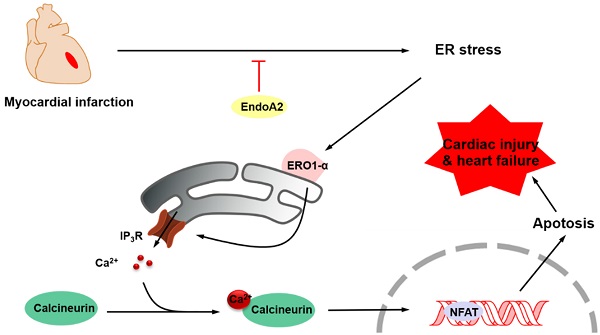
Cardiac injury upon myocardial infarction (MI) is the leading cause of heart failure. The present study aims to investigate the role of EndoA2 in ischemia-induced cardiomyocyte apoptosis and cardiac injury. In vivo, we established an MI mouse model by ligating the left anterior descending (LAD) coronary artery, and intramyocardial injection of adenoviral EndoA2 (Ad-EndoA2) was used to overexpress EndoA2. In vitro, we used the siRNA and Ad-EndoA2 transfection strategies. Here, we reported that EndoA2 expression was remarkably elevated in the infarct border zone of MI mouse hearts and neonatal rat cardiomyocytes (NRCMs) stimulated with oxygen and glucose deprivation (OGD) which mimicked ischemia. We showed that intramyocardial injection of Ad-EndoA2 attenuated cardiomyocyte apoptosis and reduced endoplasmic reticulum (ER) stress in response to MI injury. Using siRNA for knockdown and Ad-EndoA2 for overexpression, we validated that knockdown of EndoA2 in NRCMs exacerbated OGD-induced NRCM apoptosis, whereas overexpression of EndoA2 attenuates OGD-induced cardiomyocyte apoptosis. Mechanistically, knockdown of EndoA2 activated ER stress response, which increases ER oxidoreductase 1α (ERO1α) and inositol 1, 4, 5-trisphosphate receptor (IP3R) activity, thus led to increased intracellular Ca2+ accumulation, followed by elevated calcineurin activity and nuclear factor of activated T-cells (NFAT) dephosphorylation. Pretreatment with the IP3R inhibitor 2-Aminoethoxydiphenylborate (2-APB) attenuated intracellular Ca2+ accumulation, and pretreatment with the Ca2+ chelator 1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA) or the calcineurin inhibitor Cyclosporin A (CsA) inhibited EndoA2-knockdown-induced NRCM apoptosis. Overexpression of EndoA2 led to the opposite effects by suppressing ER-stress-mediated ERO1α/IP3R signaling pathway. This study demonstrated that EndoA2 protected cardiac function in response to MI via attenuating ER-stress-mediated ERO1α/IP3R signaling pathway. Targeting EndoA2 is a potential therapeutic strategy for the prevention of postinfarction-induced cardiac injury and heart failure.
Keywords: endophilin A2, cardiac injury, ER stress, ERO1α, IP3R

 Global reach, higher impact
Global reach, higher impact