Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(1):82-95. doi:10.7150/ijbs.57919 This issue Cite
Research Paper
Deoxyribonuclease 1-like 3 Inhibits Hepatocellular Carcinoma Progression by Inducing Apoptosis and Reprogramming Glucose Metabolism
1. Department of Cardiovascular Surgery, Zhongnan Hospital of Wuhan University, Wuhan 430071, P.R. China.
2. Department of Hepatobiliary Surgery, Zhongnan Hospital of Wuhan University, Wuhan 430071, P.R. China.
3. Department of Urology, Renmin Hospital of Wuhan University, Wuhan 430060, China.
4. Department of Geriatrics, Tianjin Medical University General Hospital Hospital, Tianjin, 300052, P.R. China.
5. Department of Hepatobiliary Surgery, Tianjin Medical University General Hospital , Tianjin, 300052, P.R. China.
Abstract
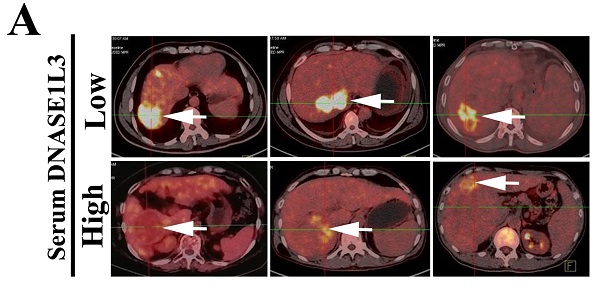
HCC has remained one of the challenging cancers to treat, owing to the paucity of drugs targeting the critical survival pathways. Considering the cancer cells are deficient in DNase activity, the increase of an autonomous apoptisis endonuclease should be a reasonable choice for cancer treatment. In this study, we investigated whether DNASE1L3, an endonuclease implicated in apoptosis, could inhibit the progress of HCC. We found DNASE1L3 was down-regulated in HCC tissues, whereas its high expression was positively associated with the favorable prognosis of patients with HCC. Besides, serum DNASE1L3 levels were lower in HCC patients than in healthy individuals. Functionally, we found that DNASE1L3 inhibited the proliferation of tumor cells by inducing G0/G1 cell cycle arrest and cell apoptosis in vitro. Additionally, DNASE1L3 overexpression suppressed tumor growth in vivo. Furthermore, we found that DNASE1L3 overexpression weakened glycolysis in HCC cells and tissues via inactivating the rate-limiting enzymes involved in PTPN2-HK2 and CEBPβ-p53-PFK1 pathways. Finally, we identified the HBx to inhibit DNASE1L3 expression by up-regulating the expression of ZNF384. Collectively, our findings demonstrated that DNASE1L3 could inhibit the HCC progression through inducing cell apoptosis and weakening glycolysis. We believe DNASE1L3 could be considered as a promising prognostic biomarker and therapeutic target for HCC.
Keywords: DNASE1L3, hepatocellular carcinoma, apoptosis, reprogramming glucose metabolism, ZNF384.

 Global reach, higher impact
Global reach, higher impact