Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(1):242-260. doi:10.7150/ijbs.66506 This issue Cite
Research Paper
Exosomal LBH inhibits epithelial-mesenchymal transition and angiogenesis in nasopharyngeal carcinoma via downregulating VEGFA signaling
1. Department of Cardiology, Laboratory of Heart Center; Guangdong Provincial Biomedical Engineering Technology Research Center for Cardiovascular Disease, Zhujiang Hospital, Southern Medical University, 253# Middle Industrial Avenue, Guangzhou, PR China, 510280.
2. Key Laboratory of Nephrology, National Health Commission and Guangdong Province; Department of Nephrology, The First Affiliated Hospital, Sun Yat-sen University, 151# Yanjiang Road, Guangzhou, PR China, 510080.
3. Nanfang Hospital, the First School of Clinical Medicine, Southern Medical University, 1023# Shatai Road South, Guangzhou, PR China, 510515.
4. State Key Laboratory of Oncology in South China; Collaborative Innovation Center for Cancer Medicine; Guangdong Key Laboratory of Nasopharyngeal Carinoma Diagnosis and Therapy, Sun Yat-sen University Cancer Center, 651# Dongfeng Road East, Guangzhou, PR China, 510060.
*These authors contributed equally to this work.
Abstract
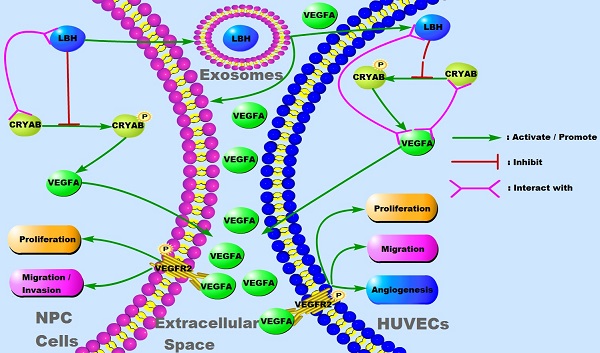
The limb-bud and heart (LBH) gene was reported to suppress nasopharyngeal carcinoma (NPC) progression in our previous study. Distant metastasis predominantly accounts for the unsatisfactory prognosis of NPC treatment, in which epithelial-mesenchymal transition (EMT) and tumor angiogenesis are of great significance. The roles of exosomes in mediating NPC progression have been highlighted in recent researches, and attempts have been made to explore the clinical application of NPC exosomes. Here we investigated the function of the LBH gene in NPC exosomes, and its potential mechanism. NPC xenografts were constructed, showing that vascular endothelial growth factor A (VEGFA) expression and neovascularity were attenuated by LBH overexpression, together with diminished EMT progression. NPC-derived exosomes were isolated, identified and applied for in vitro/in vivo experiments, and the exosomal distribution of LBH was elevated in exosomes derived from LBH-upregulated cells. Ectopic LBH, αB-crystallin (CRYAB) and VEGFA expression was induced by lentiviral infection or plasmid transfection to explore their functions in modulating EMT and angiogenesis in NPC. The addition of LBH+ NPC exosomes during a Matrigel plug assay in mice suppressed in vivo angiogenesis, and the treatment of human umbilical vein endothelial cells (HUVECs) with LBH+ NPC exosomes inhibited cellular proliferation, migration and tube formation. The interactions among LBH, CRYAB and VEGFA were confirmed by colocalization and fluorescence resonance energy transfer (FRET) assays, and extracellular VEGFA secretion from both HUVECs and NPC cells under the treatment with LBH+ NPC exosomes was diminished according to ELISA results. We concluded that exosomal LBH inhibits EMT progression and angiogenesis in the NPC microenvironment, and that its effects are partially implemented by modulation of VEGFA expression, secretion and related signaling. Thus, LBH could serve as a promising therapeutic target in VEGFA-focused NPC treatment.
Keywords: LBH, Exosomes, EMT progression, Angiogenesis, VEGFA, Nasopharyngeal carcinoma

 Global reach, higher impact
Global reach, higher impact