Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(2):652-660. doi:10.7150/ijbs.64188 This issue Cite
Research Paper
Genome Editing with AAV-BR1-CRISPR in Postnatal Mouse Brain Endothelial Cells
1. State Key Laboratory of Proteomics, Beijing Proteome Research Center, National Center for Protein Sciences (Beijing), Beijing Institute of Lifeomics, Beijing 102206, China.
2. Department of Immunology, College of Basic Medicine, Qingdao University, Qingdao, Shandong 266071, China.
Abstract
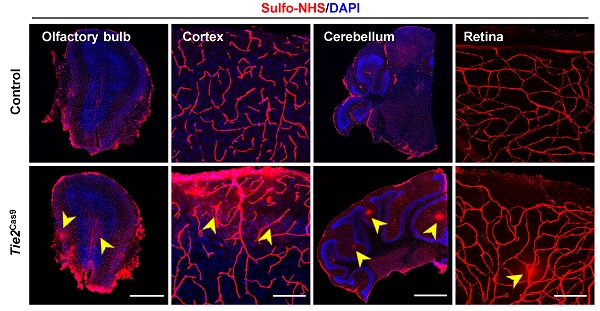
Brain endothelial cells (ECs) are an important component of the blood-brain barrier (BBB) and play key roles in restricting entrance of possible toxic components and pathogens into the brain. However, identifying endothelial genes that regulate BBB homeostasis remains a time-consuming process. Although somatic genome editing has emerged as a powerful tool for discovery of essential genes regulating tissue homeostasis, its application in brain ECs is yet to be demonstrated in vivo. Here, we used an adeno-associated virus targeting brain endothelium (AAV-BR1) combined with the CRISPR/Cas9 system (AAV-BR1-CRISPR) to specifically knock out genes of interest in brain ECs of adult mice. We first generated a mouse model expressing Cas9 in ECs (Tie2Cas9). We selected endothelial β-catenin (Ctnnb1) gene, which is essential for maintaining adult BBB integrity, as the target gene. After intravenous injection of AAV-BR1-sgCtnnb1-tdTomato in 4-week-old Tie2Cas9 transgenic mice resulted in mutation of 36.1% of the Ctnnb1 alleles, thereby leading to a dramatic decrease in the level of CTNNB1 in brain ECs. Consequently, Ctnnb1 gene editing in brain ECs resulted in BBB breakdown. Taken together, these results demonstrate that the AAV-BR1-CRISPR system is a useful tool for rapid identification of endothelial genes that regulate BBB integrity in vivo.
Keywords: CRISPR/Cas9, brain endothelial cell, blood-brain barrier, genome editing, AAV-BR1

 Global reach, higher impact
Global reach, higher impact