Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(3):956-969. doi:10.7150/ijbs.65954 This issue Cite
Research Paper
A Sonic Hedgehog-Gli-Bmi1 signaling pathway plays a critical role in p27 deficiency induced bone anabolism
1. State Key Laboratory of Reproductive Medicine; Research Center for Bone and Stem Cells; Department of Anatomy, Histology and Embryology; Key Laboratory for Aging & Disease; Nanjing Medical University, Nanjing 211166, China.
2. Calcium Research Laboratory, McGill University Health Centre and Department of Medicine, McGill University, Montreal, Quebec H3A 1A1, Canada.
3. The Research Center for Aging, Affiliated Friendship Plastic Surgery Hospital of Nanjing Medical University, Nanjing 210029, China.
Abstract
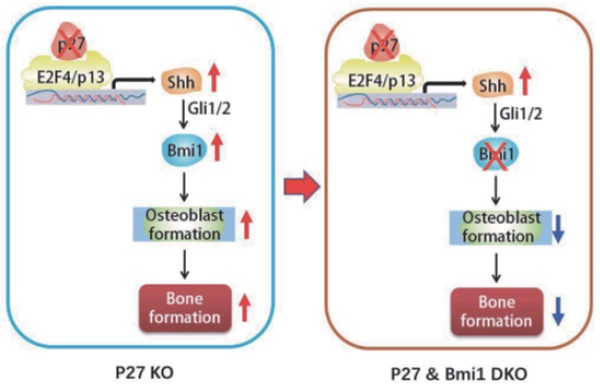
To explore the mechanism of the bone anabolic action of p27 deficiency, we first confirmed that osteoblast formation and osteogenesis were significantly increased in p27 deficient mice compared with their wild-type littermates. Microarray analysis of differential gene expression profiles, followed by real-time RT-PCR and Western blots revealed that p27 deletion significantly upregulated the expression of Sonic hedgehog (Shh), Gli1 and 2 and their target gene Bmi1 in bone tissue, and significantly down regulated the expression of the negative regulators of the Shh pathway Sufu, Patched 1 and Gli3 in bone tissue. The Shh antagonist KAAD-cyclopamine or vismodegib significantly reduced osteogenesis of bone marrow mesenchymal stem cells (BM-MSCs) in vitro and osteoblastic bone formation in vivo. The results of chromatin immunoprecipitation and double luciferase assay demonstrated that p27 inhibited Shh transcription mediated via E2F4. Bmi1 knockout blocked the increase of osteoblastic bone formation induced by p27 deficiency in vivo. In conclusion, the results of this study indicate that the signaling pathway Shh-Gli-Bmi1 plays a critical role in p27 deficiency induced bone anabolic action, suggesting that Bmi1 may be an important therapeutic target for osteoporosis induced by activation of p27 signaling or inactivation of sonic hedgehog signaling.
Keywords: p27, osteogenesis, osteoblast formation, sonic hedgehog signaling, Bmi1

 Global reach, higher impact
Global reach, higher impact