Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(3):1254-1270. doi:10.7150/ijbs.65174 This issue Cite
Research Paper
Integrin-β4 regulates the dynamic changes of phenotypic characteristics in association with epithelial-mesenchymal transition (EMT) and RhoA activity in airway epithelial cells during injury and repair
1. School of Basic Medicine, Central South University, Changsha, Hunan 410078, China
2. Changzhou Key Laboratory of Respiratory Medical Engineering, Institute of Biomedical Engineering and Health Sciences, Changzhou, Jiangsu 213164, China
3. Department of Reproductive Medicine, Liuzhou maternity and Child Healthcare Hospital, Affiliated Women and Children's Hospital of Guangxi university of Science and Technology, Liuzhou, Guangxi 545001, China
4. School of Nursing, Changzhou University, Changzhou, Jiangsu 213164, China
5. Xiangtan Central Hospital, Xiangtan, Hunan 411100, China
*These authors contributed equally to the work
Abstract
Background: In airway disease such as asthma a hyperactive cellular event of epithelial-mesenchymal transition (EMT) is considered as the mechanism of pathological airway tissue remodeling after injury to the airway epithelium. And the initiation of EMT in the airways depends on the epithelial disruption involving dissolution and/or destabilization of the adhesive structures between the cells and ECM. Previously, we have shown that integrin-β4, an epithelial adhesion molecule in bronchial epithelium is an important regulator of cell proliferation and wound repair in human airway epithelial cells. Therefore, in this study we aimed to investigate whether integrin-β4 also regulates EMT phenotypes during injury and repair in airway epithelial cells of both wild type/integrin-β4-/- mice in vivo and cultured cells treated with integrin-β4/nonsense siRNA in vitro.
Methods: We induced injury to the airway epithelial cells by either repeated exposure to ozone and mechanical scratch wound, and subsequently examined the EMT-related phenotypic features in the airway epithelial cells including biomarkers expression, adhesion and cytoskeleton reorganization and cell stiffness.
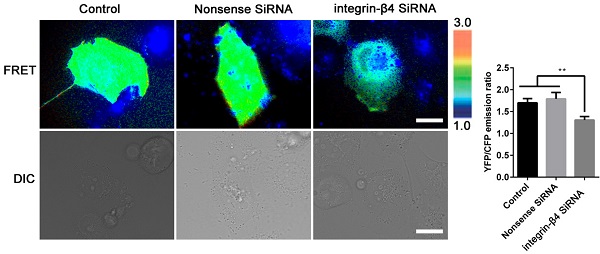
Results: The results show that in response to injury (ozone exposure/scratch wound) and subsequent spontaneous repair (ozone withdrawal/wound healing) both in vivo and in vitro, the airway epithelial cells underwent dynamic changes in the epithelial and mesenchymal biomarkers expression, adhesion and cytoskeleton structures as well as cell stiffness, all together exhibiting enhanced EMT phenotypic features after injury and reversal of the injury-induced effects during repair. Importantly, these injury/repair-associated EMT phenotypic changes in airway epithelial cells appeared to be dependent on integrin-β4 expression. More specifically, when integrin-β4 was deficient in mice (integrin-β4-/-) the repair of ozone-injured airway epithelium was impaired and the recovery of ozone-enhanced EMT biomarkers expression in the airway epithelium was delayed. Similarly, in the scratch wounded airway epithelial cells with integrin-β4 knockdown, the cells were impaired in all aspects related to EMT during wound and repair including cell proliferation, wound closure rate, adhesion and cytoskeleton protein expression (vinculin and vimentin), mesenchymal-like F-actin reorganization, cell stiffness and RhoA activation.
Conclusion: Taken together, these results suggested that integrin-β4 may be essential in regulating the effects of injury and repair on EMT in airway epithelial cells via influencing both the cell adhesion to ECM and cells' physical phenotypes through RhoA signaling pathway.
Keywords: Airway epithelium, wound repair, EMT, integrin-β4, cell stiffness

 Global reach, higher impact
Global reach, higher impact