Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(5):2186-2201. doi:10.7150/ijbs.60594 This issue Cite
Research Paper
SENP1 promotes triple-negative breast cancer invasion and metastasis via enhancing CSN5 transcription mediated by GATA1 deSUMOylation
1. Department of General Surgery, Tianjin Medical University General Hospital, Tianjin, China.
2. Department of Obstetrics and Gynecology. Tianjin Medical University General Hospital, Tianjin, China.
3. Department of Nuclear Medicine and Molecular Imaging, Tianjin Medical University Cancer Institute and Hospital, National Clinical Research Center for Cancer, Tianjin, China.
4. Department of Breast Cancer Pathology, Tianjin Medical University Cancer Institute and Hospital, National Clinical Research Center for Cancer, Tianjin, China.
5. The Second Surgical Department of Breast Cancer Oncology, Tianjin Medical University Cancer Institute and Hospital, Key Laboratory of Breast Cancer Prevention and Therapy, National Clinical Research Center for Cancer, Tianjin, China.
Abstract
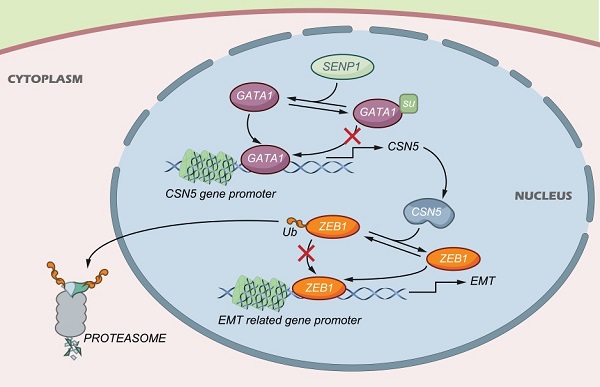
TNBC is characterized by high incidence of visceral metastasis and lacks effective clinical targets. This study aims to delineate the molecular mechanisms of SENP1 in TNBC invasion and metastasis. By using IHC to test the SENP1 expression in TNBC tissues, we analyzed the relationship between SENP1 expression and TNBC prognosis. We showed that SENP1 expression was higher in TNBC tumor tissues and related to TNBC prognosis, supporting SENP1 as an independent risk factor. High expression of SENP1 was significantly associated with histologic grade and tumor lymph node invasion. Intriguingly, the expression levels of SENP1 in TNBC tumors were significantly correlated with that of CSN5, GATA1 and ZEB1. Importantly, SENP1 promoted TNBC cell migration and invasion by regulating ZEB1 deubiquitination and expression through CSN5. Further studies showed that deSUMOylation at lysine residue K137 of GATA1 enhanced the binding of GATA1 to the CSN5 promoter and transactivated CSN5 expression. In addition, we showed that ZEB1 is deubiquitinated at lysine residue K1108. Our in vivo studies also indicated that reduction in SENP1 expression upregulated GATA1 SUMOylation, and thus resulted in decreased expression of CSN5 and ZEB1 in the tumor microenvironment, which decelerated TNBC progression and metastasis. SENP1 promoted CSN5-mediated ZEB1 protein degradation via deSUMOylation of GATA1, and thus influenced TNBC progression. These findings suggest that SENP1 could be utilized as a potential target for blockade of TNBC development and thus provide a totally new approach for TNBC treatment.
Keywords: Triple-negative breast cancer, SENP-1, deSUMOylation, GATA-1, CSN5, ZEB1

 Global reach, higher impact
Global reach, higher impact