Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(7):2795-2806. doi:10.7150/ijbs.71595 This issue Cite
Review
Smad3 Signatures in Renal Inflammation and Fibrosis
1. Guangdong-Hong Kong Joint Laboratory for Immunological and Genetic Kidney Disease, Department of Pathology, and Guangdong Cardiovascular Institute, Guangdong Academy of Medical Sciences, Guangdong Provincial People's Hospital, Guangzhou, China.
2. Departments of Medicine & Therapeutics, Li Ka Shing Institute of Health Sciences, and Lui Che Woo Institute of Innovative Medicine, The Chinese University of Hong Kong, Hong Kong, China.
3. Hubei University of Chinese Medicine, Wuhan, China.
4. Department of Nephrology, Hubei Provincial Hospital of Traditional Chinese Medicine, Hubei Province Academy of Traditional Chinese Medicine, Wuhan, China.
5. The Chinese University of Hong Kong-Guangdong Academy of Sciences/ Guangdong Provincial People's Hospital Joint Research Laboratory on Immunological and Genetic Kidney Diseases, The Chinese University of Hong Kong, Hong Kong, China.
Abstract
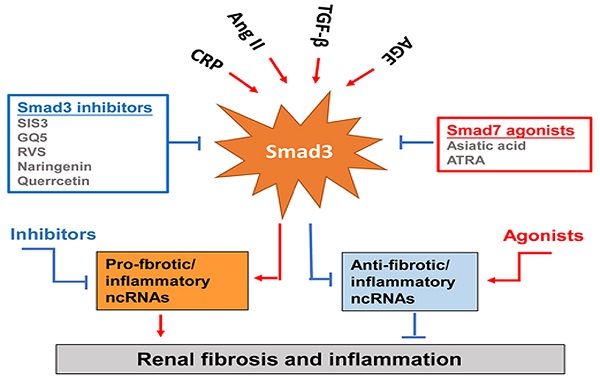
Renal inflammation and fibrosis are key pathological features of acute kidney injury (AKI) and chronic kidney disease (CKD). Smad3 is a critical mediator of TGF-β signaling and plays a pathogenic role in both renal inflammation and fibrosis. Smad3 can be activated not only by TGF-β1 but also by many stress molecules including angiotensin II (Ang II), advanced end products (AGEs), and C-reactive protein (CRP) under disease conditions. In addition, Smad3 can interact with other signaling pathways, such as the ERK/p38 MAPK and NF-κB pathways, to mediate renal inflammation and fibrosis. Mechanistically, Smad3 transcriptionally regulates many downstream target genes including microRNAs and long non-coding RNAs to cause cell death, inflammation, and fibrosis. Thus, targeting Smad3 or its downstream genes specifically related to renal inflammation and fibrosis should provide a novel therapeutic strategy to combat kidney diseases.
Keywords: Smad3, renal inflammation and fibrosis, miRNAs, lncRNAs

 Global reach, higher impact
Global reach, higher impact