Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(7):2882-2897. doi:10.7150/ijbs.69655 This issue Cite
Research Paper
CIRBP-OGFR axis safeguards against cardiomyocyte apoptosis and cardiotoxicity induced by chemotherapy
1. Department of Biochemistry and Molecular Biology, Beijing Key Laboratory of Protein Posttranslational Modifications and Cell Function, School of Basic Medical Sciences, Peking University Health Science Center, 38 Xueyuan Road, Beijing 100191, China.
2. National Health Commission Key Laboratory of Cardiovascular Regenerative Medicine, Heart Center of Henan Provincial People's Hospital, Central China Fuwai Hospital of Zhengzhou University, Fuwai Central China Cardiovascular Hospital & Central China Branch of National Center for Cardiovascular Diseases, Zhengzhou, Henan, 451464, China.
3. Department of Children's Heart Center, Henan Provincial People's Hospital, Department of Children's Heart Center of Central China Fuwai Hospital, Henan Key Medical Laboratory of Tertiary Prevention and Treatment for Congenital Heart Disease, Central China Fuwai Hospital of Zhengzhou University, Zhengzhou, Henan, 451464, China.
4. Henan Key Laboratory of Chronic Disease Management, Department of Health Management Center, Henan Provincial People's Hospital, Department of Health Management Center of Central China Fuwai Hospital, Central China Fuwai Hospital of Zhengzhou University, Zhengzhou, Henan, 451464, China.
5. Department of Cardiology, Henan Provincial People's Hospital, Department of Cardiology of Central China Fuwai Hospital, Henan Key Laboratory for Coronary Heart Disease Prevention and Control, Central China Fuwai Hospital of Zhengzhou University, Zhengzhou, Henan, 451464, China.
6. State Key Laboratory of Cardiovascular Disease, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100037, China.
7. Department of Hematology, Henan Provincial People's Hospital, Zhengzhou, Henan, 450003, China.
8. Henan Key Laboratory of Molecular Pathology, Department of Molecular Pathology, The Affiliated Cancer Hospital of Zhengzhou University, Zhengzhou, Henan, 450008, China.
#These authors contributed equally to this work and should be considered as co-first authors.
Abstract
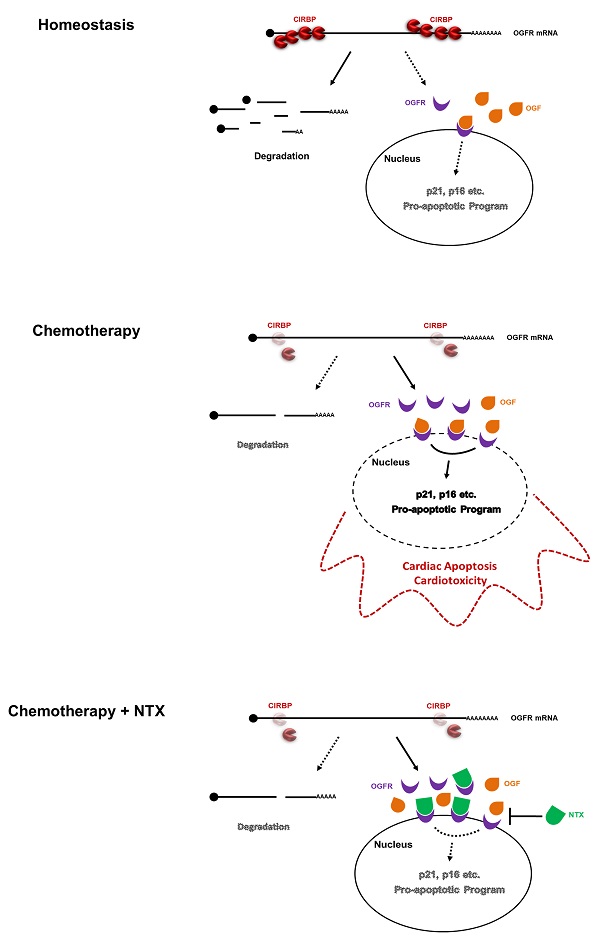
Cold-inducible RNA-binding protein (CIRBP) is documented to be required for maintaining cardiac function, however, its role in chemotherapy-induced cardiotoxicity remains obscured. Herein, we report that CIRBP decreases cardiomyocyte apoptosis and attenuates cardiotoxicity through disrupting OGF-OGFR signal. CIRBP deficiency is involved in diverse chemotherapeutic agents induced cardiomyocyte apoptosis. Delivery of exogenous CIRBP to the mouse myocardium significantly mitigated doxorubicin-induced cardiac apoptosis and dysfunction. Specifically, OGFR was identified as a downstream core effector responsible for chemotherapy-induced cardiomyocyte apoptosis. CIRBP was shown to interact with OGFR mRNA and to repress OGFR expression by reducing mRNA stability. CIRBP-mediated cytoprotection against doxorubicin-induced cardiac apoptosis was demonstrated to largely involve OGFR repression by CIRBP. NTX as a potent antagonist of OGFR successfully rescued CIRBP ablation-rendered susceptibility to cardiac dyshomeostasis upon exposure to doxorubicin, whereas another antagonist ALV acting only on opioid receptors did not. Taken together, our results demonstrate that CIRBP confers myocardium resistance to chemotherapy-induced cardiac apoptosis and dysfunction by dampening OGF/OGFR axis, shedding new light on the mechanisms of chemo-induced cardiotoxicity and providing insights into the development of an efficacious cardioprotective strategy for cancer patients.
Keywords: CIRBP, Cardioprotection, OGFR, Chemotherapy-induced cardiotoxicity, Cardiomyocyte apoptosis

 Global reach, higher impact
Global reach, higher impact