Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(10):4245-4259. doi:10.7150/ijbs.72204 This issue Cite
Research Paper
RASAL2 mediated the enhancement of YAP1/TIAM1 signaling promotes malignant phenotypes of pancreatic ductal adenocarcinoma
1. Department of Hepatobiliary Surgery, First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi 710061, China.
2. Department of Vascular Surgery, First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi 710061, China.
3. Department of Urology Surgery, First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi 710061, China.
4. Department of Ophthalmology, First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi 710061, China.
5. Center for Pancreatic Cancer Research, South China University of Technology, Guangzhou, Guangdong 510006, China.
*These authors contributed equally to this work.
Abstract
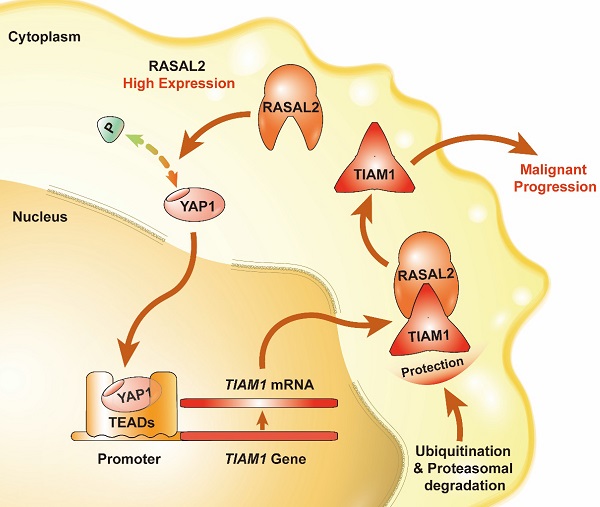
Pancreatic ductal adenocarcinoma (PDAC) is characterized by a high incidence of metastasis and dismal prognosis. As a member of Gas-Gap gene, RASAL2 is involved in the hydrolysis of RAS-GTP to RAS-GDP and abnormal expression in human cancers. Here we firstly described the function of RASAL2 on PDAC to enrich the knowledge of RAS family.We interestingly observed that RASAL2 expression was upregulated in PDAC at both mRNA and protein levels, and high expression of RASAL2 predicted a poor prognosis in PDAC patients. Additionally, RASAL2 promoted malignant behaviors of PDAC in vitro and in vivo. To determine the mechanistic roles of RASAL2 signaling and its potential as a therapeutic target in PDAC, we clarified that RASAL2 could accumulate the TIAM1 expression in different level through inhibiting YAP1 phosphorylation, increased TIAM1 mRNA expression and suppressed ubiquitination of TIAM1 protein. In conclusion, RASAL2 enhances YAP1/TIAM1 signaling and promotes PDAC development and progression.
Keywords: RASAL2, YAP1, TIAM1, PDAC, progression

 Global reach, higher impact
Global reach, higher impact