Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(11):4357-4371. doi:10.7150/ijbs.69969 This issue Cite
Research Paper
Genome-Wide CRISPR/Cas9 Library Screening Identified that DUSP4 Deficiency Induces Lenvatinib Resistance in Hepatocellular Carcinoma
1. Department of General Surgery, Guangdong Provincial People's Hospital, Guangdong Academy of Medical Sciences, Guangzhou 510080, China
2. Heyuan people's Hospital, Heyuan 517000, China
3. School of Medicine, South China University of Technology, Guangzhou 510080, China
4. The Second School of Clinical Medicine, Southern Medical University, Guangzhou, 510515, China
5. Shantou University of Medical College, Shantou, 515000, China
6. Department of General Surgery, Hui Ya Hospital of The First Affiliated Hospital of Sun Yat-Sen University; Department of Liver Surgery, The First Affiliated Hospital of Sun Yat-Sen University, Guangzhou, 510080, China
7. Department of Hepatobiliary Surgery, the Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, 510095, China
8. Organ Transplant Centre, The First Affiliated Hospital, Sun Yat-sen University, Guangdong Provincial Key Laboratory of Organ Donation and Transplant Immunology, Guangdong Provincial International Cooperation Base of Science and Technology (Organ Transplantation), Guangzhou 510080, China
*The authors contributed equally to this manuscript and should all be considered as co-first authors.
Abstract
Background: Lenvatinib is in a first-line therapy for advanced hepatocellular carcinoma (HCC). However, drug resistance is one of the principal obstacles for treatment failure. The molecular mechanism of Lenvatinib resistance has not been well investigated.
Materials and methods: A genome-wide CRISPR/Cas9 knockout screening system was established and bioinformatic analysis was used to identify critical genes associated with Lenvatinib resistance. Cell proliferation assays, colony formation assays and cell migration assays were performed to investigate the effect of drug resistance associated genes, particularly DUSP4, on cancer cell malignant behavior during Lenvatinib treatment. In vivo experiments were conducted by using a xenograft mouse model.
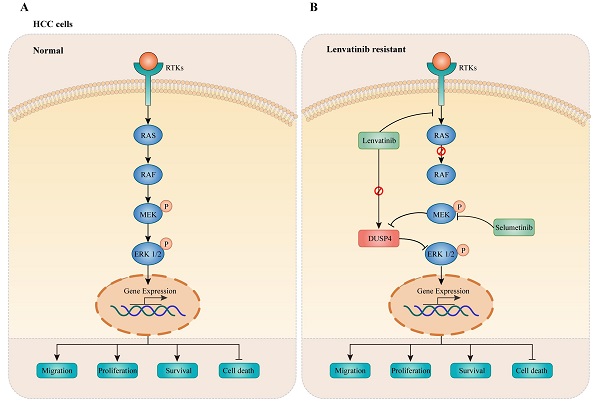
Results: We identified six genes that were associated with Lenvatinib resistance in HCC, including DUSP4, CCBL1, DHDH, CNTN2, NOS3 and TNF. DUSP4 was found to be significantly decreased at the mRNA and protein levels in Lenvatinib resistant HCC cells. DUSP4 knockout enhanced HCC cell survival, cell proliferation and migration during Lenvatinib treatment in vitro and in vivo, accompanied by regulation of p-ERK and p-MEK levels. This finding implied that DUSP4 deficiency induced Lenvatinib resistance. Interestingly, DUSP4 deficiency induced Lenvatinib resistance was abrogated by the MEK inhibitor Selumetinib, implying that MEK phosphorylation and DUSP4-inhibition dependent ERK activation were required for drug resistance. Finally, we found that DUSP4 deficiency was associated with HCC prognosis and response to Lenvatinib based on clinical data.
Conclusions: DUSP4 deficiency mediates Lenvatinib resistance by activating MAPK/ERK signaling and combination therapy using Lenvatinib and MEK inhibitors may be a promising therapeutic strategy for overcoming Lenvatinib resistance.
Keywords: Lenvatinib, drug resistance, genome-wide CRISPR/Cas9 library, DUSP4, MAPK/ERK pathway

 Global reach, higher impact
Global reach, higher impact