Impact Factor ISSN: 1449-2288
Int J Biol Sci 2023; 19(3):865-879. doi:10.7150/ijbs.79355 This issue Cite
Research Paper
G protein inhibitory α subunit 2 is a molecular oncotarget of human glioma
1. Institute of Neuroscience, Soochow University, Institute for Excellence in Clinical Medicine of Kunshan First People's Hospital and Soochow University, Suzhou, China.
2. Clinical Research Center of Neurological Disease, The Second Affiliated Hospital of Soochow University, Jiangsu Key Laboratory of Neuropsychiatric Diseases and Institute of Neuroscience, Soochow University, Suzhou, China.
3. Department of Neurosurgery, The affiliated Changzhou No.2 People's Hospital of Nanjing Medical University, Changzhou, China.
4. Department of Neurosurgery, the First Affiliated Hospital of Soochow University, Suzhou, China.
5. The Affiliated Eye Hospital, Nanjing Medical University, Nanjing, China.
#Co-first authors.
Abstract
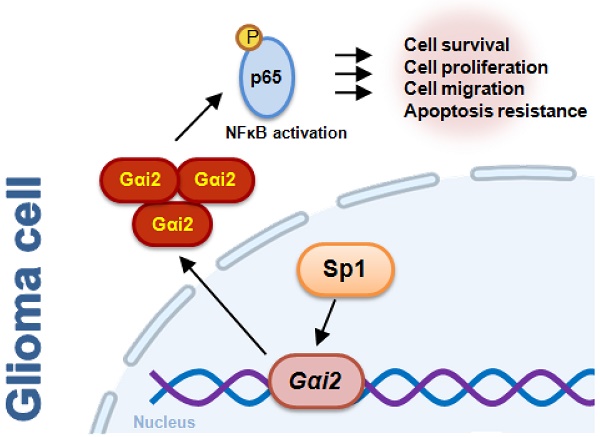
Identification of novel therapeutic oncotargets for human glioma is extremely important. Here we tested expression, potential functions and underlying mechanisms of G protein inhibitory α subunit 2 (Gαi2) in glioma. Bioinformatics analyses revealed that Gαi2 expression is significantly elevated in human glioma, correlating with poor patients' survival, higher tumor grade and wild-type IDH status. Moreover, increased Gαi2 expression was also in local glioma tissues and different glioma cells. In primary and immortalized (A172) glioma cells, Gαi2 shRNA or knockout (KO, by Cas9-sgRNA) potently suppressed viability, proliferation, and mobility, and induced apoptosis. Ectopic Gαi2 overexpression, using a lentiviral construct, further augmented malignant behaviors in glioma cells. p65 phosphorylation, NFκB activity and expression of NFκB pathway genes were decreased in Gαi2-depleted primary glioma cells, but increased following Gαi2 overexpression. There was an increased binding between Gαi2 promoter and Sp1 (specificity protein 1) transcription factor in glioma tissues and different glioma cells. In primary glioma cells Gαi2 expression was significantly reduced following Sp1 silencing, KO or inhibition. In vivo studies revealed that Gαi2 shRNA-expressing AAV intratumoral injection hindered growth of subcutaneous glioma xenografts in nude mice. Moreover, Gαi2 KO inhibited intracranial glioma xenograft in nude mice. Gαi2 depletion, NFκB inhibition and apoptosis induction were observed in subcutaneous and intracranial glioma xenografts with Gαi2 depletion. Together, overexpressed Gαi2 is important for glioma cell growth possibly by promoting NFκB cascade activation.

 Global reach, higher impact
Global reach, higher impact