10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2023; 19(9):2934-2956. doi:10.7150/ijbs.81961 This issue Cite
Research Paper
Colchicine attenuates the electrical remodeling of post-operative atrial fibrillation through inhibited expression of immune-related hub genes and stabilization of microtubules
1. Department of Cardiology, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou 310016, China.
2. Key Laboratory of Cardiovascular Intervention and Regenerative Medicine of Zhejiang Province, Hangzhou 310016, China.
3. Department of Breast Surgery, Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310009, China.
4. Key Laboratory of Tumor Microenvironment and Immune Therapy of Zhejiang Province, Hangzhou 310009, China.
5. Key Laboratory of combined Multi-organ Transplantation, Ministry of Public Health, First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310003, China.
6. Institute of Translational Medicine, Zhejiang University, Hangzhou 310029, China.
#These authors contributed equally
Abstract
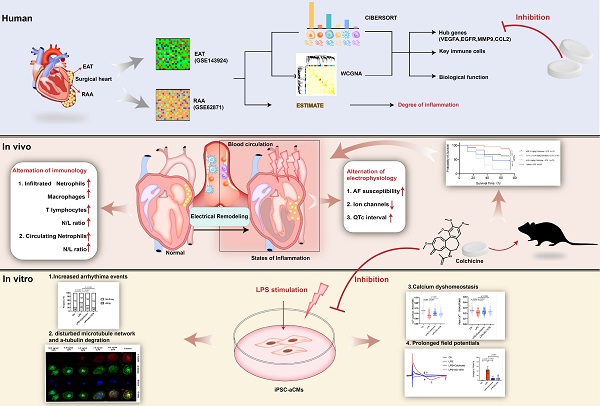
Rationale: Acute inflammation is a major risk factor for post-operative atrial fibrillation (POAF), and epicardial adipose tissue (EAT) is considered as a source of inflammatory mediators. However, underlying mechanisms and pharmacological targets of POAF are poorly understood.
Methods: Integrative analysis of array data from EAT and right atrial appendage (RAA) samples was conducted to identify potential hub genes. Lipopolysaccharide (LPS)-stimulated inflammatory models in mice and in induced pluripotent stem cell-derived atrial cardiomyocytes (iPSC-aCMs) were used to examine the exact mechanism underlying POAF. Electrophysiological analysis, multi-electrode array, and Ca2+ imaging was employed to explore the alterations of electrophysiology and Ca2+ homeostasis under inflammation. Flow cytometry analysis, histology and immunochemistry were performed to investigate immunological alterations.
Results: We observed electrical remodeling, enhanced atrial fibrillation (AF) susceptibility, immune cell activation, inflammatory infiltration, and fibrosis in LPS-stimulated mice. LPS-stimulated iPSC-aCMs showed arrhythmias, abnormal Ca2+ signaling, reduced cell viability, disrupted microtubule network and increased α-tubulin degradation. VEGFA, EGFR, MMP9 and CCL2 were identified as hub genes simultaneously targeted in the EAT and RAA of POAF patients. Notably, treatment of colchicine in LPS-stimulated mice resulted in a U-shape dose-response curve, where greatly improved survival rates were observed only at doses between 0.10-0.40 mg/kg. At this therapeutic dose level, colchicine inhibited the expression of all the identified hub genes and effectively rescued the pathogenic phenotypes observed in LPS-stimulated mice and iPSC-aCM models.
Conclusions: Acute inflammation promotes α-tubulin degradation, induces electrical remodeling, and both recruits and facilitates the infiltration of circulating myeloid cells. A certain dose of colchicine attenuates electrical remodeling and decreases the recurrence of AF.
Keywords: colchicine, post-operative atrial fibrillation, epicardial adipose tissue, bioinformatics, hub genes

 Global reach, higher impact
Global reach, higher impact