Impact Factor ISSN: 1449-2288
Int J Biol Sci 2026; 22(8):4059-4073. doi:10.7150/ijbs.130025 This issue Cite
Review
Intestinal Ischemia/Reperfusion Injury: Mechanisms, Diagnosis, and Therapeutic Advances
Department of Anesthesiology, Nanfang Hospital, Southern Medical University, Guangzhou, Guangdong, China.
# These authors contributed equally to this study.
Received 2025-12-16; Accepted 2026-2-14; Published 2026-3-30
Abstract
Intestinal ischemia/reperfusion (I/R) injury is a critical clinical syndrome precipitated by the restoration of blood flow following intestinal ischemia, a common occurrence in perioperative settings such as abdominal aortic surgery, hemorrhagic shock, and cardiopulmonary bypass. This injury extends beyond the local gut, as the disruption of the intestinal mucosal barrier facilitates bacterial and endotoxin translocation into the systemic circulation, triggering a systemic inflammatory response that can progress to sepsis and multiple organ dysfunction syndrome (MODS). Despite advances in critical care, the mortality rate associated with severe intestinal I/R injury remains formidable. The microcirculatory disturbances and organ damage following intestinal I/R involve complex pathological processes, including metabolic injury and oxidative stress. In recent years, rapid developments in the understanding of cell death mechanisms, gut microbiota, microRNAs, and fundamental medical technologies have significantly advanced research on the prevention and treatment of I/R injury. This review aims to comprehensively summarize the occurrence and progression of intestinal I/R, its impact on extraintestinal organ injury, diagnostic strategies and biomarkers, as well as current treatment methods, thereby providing guidance for the future prediction, diagnosis, and treatment of intestinal I/R injury.
Keywords: intestinal ischemia-reperfusion, extraintestinal organs injury, diagnostic strategies, biomarkers, therapeutic approaches
Introduction
Intestinal ischemia/reperfusion (I/R) injury frequently arises in perioperative settings, particularly following high-risk procedures such as severe trauma, hemorrhagic shock, severe infection, intestinal obstruction, small bowel transplantation, abdominal aortic surgery, and cardiopulmonary bypass. In such clinical scenarios, intestinal I/R injury substantially contributes to patient morbidity and mortality[1]. Despite progress in modern medical technology, the in-hospital mortality rate associated with I/R injury remains alarmingly high, ranging from 60% to 80%[2, 3], and its incidence continues to increase[3, 4]. Emerging evidence suggests that susceptibility to intestinal I/R injury may be influenced by biological sex, with females exhibiting a relatively greater tolerance than males[5]. At the core of intestinal I/R pathophysiology lies profound microcirculatory dysfunction, characterized by impaired nutrient perfusion, endothelial and epithelial injury, inflammatory cell activation, and loss of epithelial barrier integrity[6]. Importantly, microcirculatory failure represents the initiating event that converts a localized ischemic insult into a systemic inflammatory disorder. During ischemia, metabolic derangements and the accumulation of toxic metabolites prime the tissue for injury. Upon reperfusion, the abrupt restoration of blood flow triggers excessive production of reactive oxygen species (ROS) and inflammatory mediators, leading to epithelial cell death and breakdown of the intestinal barrier. This disruption facilitates the translocation of gut-derived bacteria, endotoxins, and damage-associated molecular patterns (DAMPs) into the systemic circulation and mesenteric lymph. Through this gut-centered inflammatory amplification axis, intestinal I/R injury propagates systemic immune activation, distant organ inflammation, and ultimately multiple organ dysfunction or failure[7-9]. Thus, intestinal I/R injury serves as a critical mechanistic link between localized ischemic stress and systemic inflammatory syndromes, highlighting the intestine as a central driver rather than a passive victim of multi-organ injury.
In recent years, rapid advances in the understanding of cell death mechanisms, gut microbiota, microRNA biology, and related biomedical technologies have spurred numerous innovative studies aimed at preventing or mitigating I/R injury, many of which have been published in high-impact journals. This review aims to comprehensively summarize current knowledge on the pathogenesis and progression of intestinal I/R injury, its systemic effects on distant organs, available diagnostic strategies and biomarkers, as well as existing and emerging therapeutic approaches. Ultimately, this synthesis aims to provide a foundation for improving the prediction, diagnosis, and clinical management of intestinal I/R injury.
Pathophysiology of intestinal I/R-induced intestinal injury
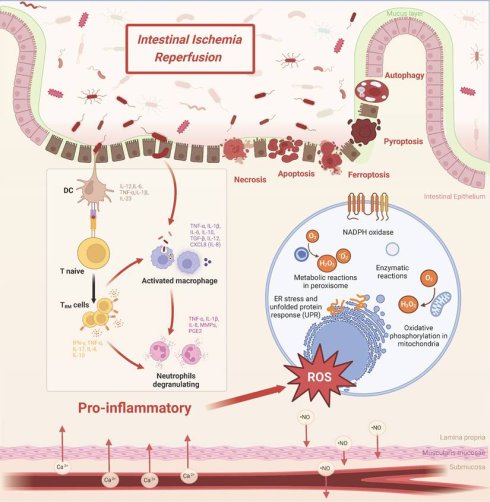
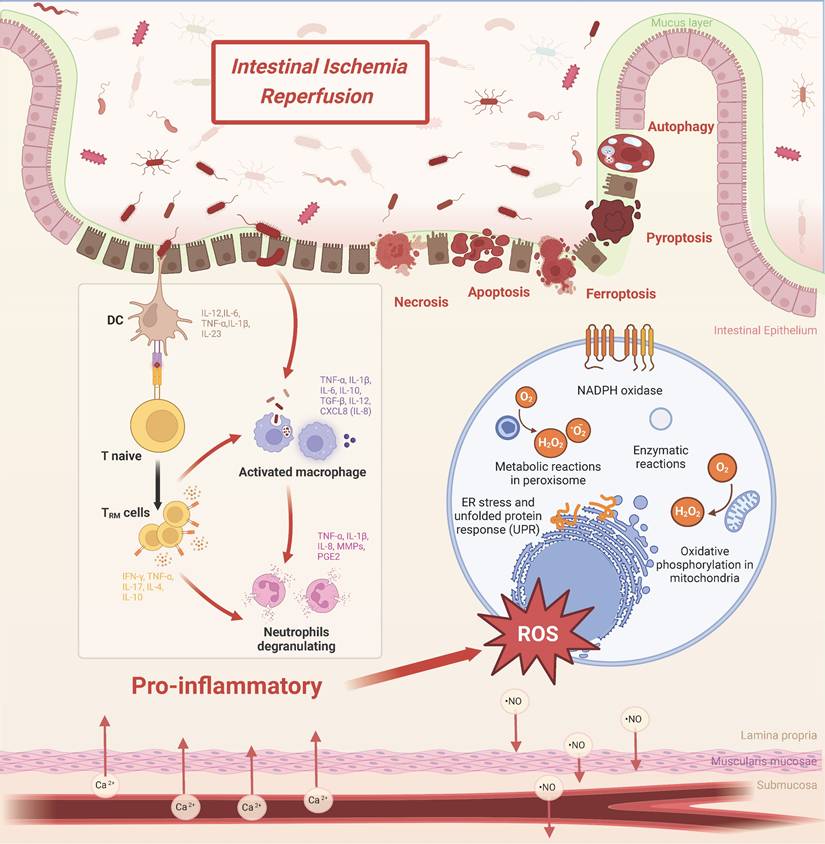
The core pathophysiology of intestinal I/R injury lies in the sequential collapse of multi-layered defense barriers. This directly leads to uncontrolled systemic inflammation. This entire process is not a singular event but rather a stereoscopic, network-like cascade initiated by ischemia and amplified upon reperfusion (Figure 1). The primary pathological manifestation is the comprehensive disruption of intestinal barrier function, encompassing the mechanical, chemical, microbial, and immune components. This breach facilitates the translocation of luminal bacteria and endotoxins, thereby driving systemic inflammatory response syndrome (SIRS) and multiple organ dysfunction[10-12]. Therefore, deciphering this intricate network is essential for developing effective therapeutic strategies.
Pathophysiological mechanisms of intestinal ischemia-reperfusion (I/R) injury. Hypoxia-reoxygenation cycles drive oxidative stress through NADPH oxidase activation, mitochondrial dysfunction, and endoplasmic reticulum (ER) stress. Immune activation involves coordinated infiltration of macrophages (releasing IL-6, TNF-α, and matrix metalloproteinases), neutrophils (producing reactive oxygen species and nitric oxide), and tissue-resident memory T cells (TRM; IL-17/IFN-γ), perpetuating mucosal inflammation. Barrier failure mechanisms arise from convergent cell death pathways: necrosis (with release of HMGB1 and ATP), NOX2-mediated apoptosis, NLRP3/gasdermin D-dependent pyroptosis, and ferroptosis driven by glutathione peroxidase 4 deficiency. Structural deterioration is characterized by depletion of goblet cells and disruption of the mucus layer.
Breach of the intestinal barrier: from structural destruction to ecological collapse
The integrity of the intestinal barrier serves as the primary line of defense in maintaining internal homeostasis, which is subjected to multi-dimensional assault during I/R injury.
Direct destruction of the mechanical and chemical Barriers
Intestinal epithelial cells, the core of the physical barrier, are primary targets of I/R injury. Ischemia directly induces a cellular energy crisis and acidosis, initiating the injury program. The subsequent burst of ROS upon reperfusion delivers the decisive blow[13-15]. This not only directly damages cellular structures but, more critically, triggers various regulated cell death pathways. These pathways include apoptosis, necroptosis, pyroptosis, ferroptosis, and autophagy[16-23]. Ultimately, this leads to extensive epithelial sloughing. Concurrently, the secretion of the protective mucus layer is reduced, and the production of chemical barrier components such as antimicrobial peptides and digestive enzymes is impaired[24-26], rendering the exposed mucosa more vulnerable to attack.
Dysbiosis of the microbial barrier: from symbiotic partner to injury accomplice
A healthy gut microbiota constitutes a vital biological barrier. The hypoxic and inflammatory milieu created by I/R rapidly induces gut dysbiosis. Its characteristic is a decrease in beneficial bacteria and an overgrowth of potentially pathogenic ones[27, 28]. This shift is not merely a passive consequence but actively amplifies injury as an amplifier. For instance, succinate produced by dysbiotic microbiota can exacerbate remote lung injury[29], while specific probiotic strains (e.g., L. murinus) or their beneficial metabolites (e.g., capsiate, CAT) can exert protective effects by inducing IL-10 secretion[30] or upregulating glutathione peroxidase 4 (GPX4) to inhibit ferroptosis[21]. Furthermore, gut microbiota-derived arginine metabolism has also been shown to mitigate intestinal I/R injury[31]. This highlights the gut microbiota as a crucial and modulable target.
Hyperactivation of the immune barrier: from defensive guard to inflammatory storm
The intestinal immune system is dramatically activated during I/R. DAMPs released from injured epithelium and invading microbial products, through pattern recognition receptors such as Toll-like receptors (TLRs)—particularly the TLR4/MyD88 axis—ignite the innate immune response[32-34]. This leads to massive infiltration of neutrophils and macrophages, which release potent weapons including neutrophil extracellular traps (NETs), causing severe “collateral damage” while attempting to clear threats[35-39]. Notably, certain immune cell subsets (e.g., ILC3s) can also secrete cytokines like IL-22 to attempt repair, underscoring the complexity of the immune response[40, 41].
Core signaling pathways: regulatory hubs of the injury network
The aforementioned barrier disruption processes are precisely orchestrated by several highly conserved core signaling pathways, which constitute promising molecular targets for intervention. Among them, the pro-inflammatory MAPK and NF-κB pathways, together with the antioxidant nuclear factor erythroid 2-related factor 2 (Nrf2) pathway, form a central regulatory network that determines cellular fate.
Key signaling pathways governing inflammation, death, and defense
The p38 MAPK/JNK pathways play a central role in stress response and cell death, and their inhibitors can significantly attenuate epithelial apoptosis[42, 43]. STING deficiency has been shown to alleviate I/R injury via JAK2/STAT3-mediated macrophage polarization and autophagy[44]. Studies indicate that the p38 MAPK inhibitor SB239063 mitigates lung injury by decreasing AQP4 levels[45]. Anesthetic agents such as propofol and dexmedetomidine also protect against I/R injury by inhibiting mitochondrial apoptosis and inflammation mediated by this pathway[46, 47]. Recent research further demonstrates that dexmedetomidine-loaded polypeptide nanospheres (PNM@Dex) alleviate intestinal I/R injury by modulating autophagy and inflammation[48]. In parallel, NF-κB acts as the master switch for pro-inflammatory gene expression, and its hyperactivation drives the production of inflammatory mediators such as TNF-α and IL-1β[49-53]. These two pathways often form positive feedback loops, mutually exacerbating the injury. Counteracting the pro-inflammatory pathways, the Nrf2 pathway serves as the central hub for cellular antioxidant defense[54]. Its activation upregulates a suite of cytoprotective proteins to combat ROS insult. For example, isoquercitrin mitigates intestinal I/R injury by regulating intestinal flora and activating the Nrf2/HO-1 signaling axis[55]. The significantly aggravated injury observed in Nrf2-knockout animals following I/R confirms its critical protective role[56].
TLR signaling: the upstream gatekeeper
The activation of these core pathways is often initiated by upstream pattern recognition receptors, among which the TLR family is particularly critical. TLR4 serves as a primary sensor for endogenous DAMPs and exogenous lipopolysaccharide (LPS). It transduces signals via adaptor proteins such as MyD88, thereby activating downstream NF-κB and MAPK pathways. This process ultimately launches the inflammatory cascade[57]. Modulating TLR4 signaling (e.g., with melatonin or shRNA-mediated silencing[39]) has proven effective in mitigating injury. Intriguingly, TLR2 signaling may, in specific contexts, exert protective effects by inducing anti-inflammatory factors like IL-10[58], suggesting context-dependent functionalities among TLR family members.
In summary, intestinal I/R injury originates from epithelial metabolic and oxidative crises. This progresses through cell death, microbial dysbiosis, and immune hyperactivation, culminating in the complete loss of the physical and functional integrity of the intestinal barrier. Intestinal barrier dysfunction and immune hyperactivation are not independent processes. Instead, they interact within a self-reinforcing pathological cycle. This cycle is initiated by oxidative stress, further amplified through inflammation driven by the TLR/NF-κB pathway, and exacerbated by the suppression of protective mechanisms such as the Nrf2 pathway. Together, these interactions create a progressively worsening pathological network. Therefore, preserving the intestinal barrier and modulating the core signaling pathways represent the key strategic approaches to interrupt this lethal cascade.
Intestinal I/R-induced extraintestinal organ injury
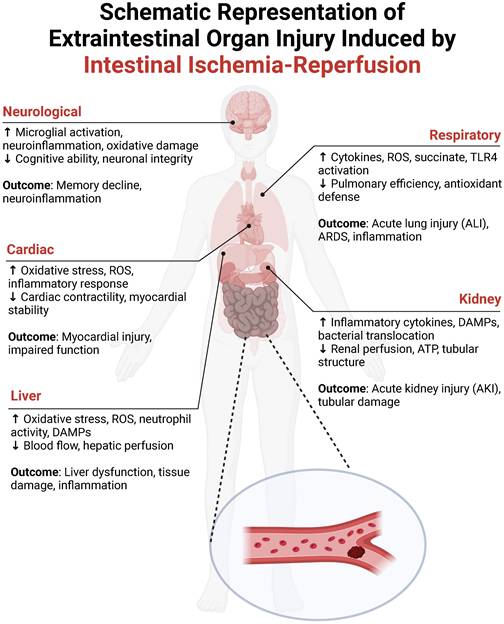
Figure 2 illustrates the systemic communication between the intestine and distant organs during I/R injury, outlining the key pathophysiological mechanisms and their consequences. The ischemic gut generates a substantial amount of ROS, which, along with a surge of inflammatory mediators (e.g., TNF-α, IL-6), enters the circulation. This “toxic effluent” can damage pulmonary vasculature and alveoli (leading to acute lung injury), constrict renal vessels and harm tubular epithelium (causing acute kidney injury), alter myocardial electrophysiology and contractility (inducing cardiac dysfunction), promote hepatocyte death (disrupting liver metabolism), and compromise the blood-brain barrier (resulting in cerebral edema and neuronal damage). Thus, intestinal I/R is a potent driver of multi-organ dysfunction, underscoring the need for a mechanistic understanding to guide clinical intervention.
Schematic representation of multiorgan injury following intestinal ischemia-reperfusion (I/R). This figure delineates systemic pathophysiology triggered by intestinal I/R. Neurological system: Neuroinflammation (microglial activation, oxidative stress) impairs neuronal integrity and cognitive function. Cardiac system: Oxidative stress and reactive oxygen species (ROS)-driven inflammation induce myocardial dysfunction and contractile impairment. Hepatic system: ROS-mediated damage, neutrophil activity, and ischemic hypoperfusion drive hepatocellular necrosis and inflammation. Respiratory system: Succinate accumulation and TLR4/ROS signaling promote acute lung injury/acute respiratory distress syndrome (ALI/ARDS) through cytokine storm and alveolar damage. Renal system: Damage-associated molecular patterns (DAMPs)-mediated inflammation and hypoperfusion trigger acute kidney injury (AKI) with characteristic tubular necrosis.
Intestinal I/R and liver injury
The intimate anatomical and functional link between the intestine and liver via the portal system makes the liver a primary target for gut-derived insults. Intestinal I/R can reduce hepatic perfusion by up to 80%[59], contributing directly to acute hepatic dysfunction[60]. More critically, the breach of the intestinal barrier allows microbial products (e.g., LPS) and DAMPs (e.g., HMGB1, histones) to translocate via the portal vein to the liver[61-64]. This influx activates hepatic innate immunity, notably via TLR4, leading to neutrophil recruitment. Neutrophils exacerbate injury through ROS release, protease secretion, and NETosis[63, 65]. The resulting sterile inflammation, combined with direct oxidative stress from systemic ROS[66, 67], drives hepatocyte apoptosis and necrosis, compromising the liver's vital metabolic and detoxifying functions.
Intestinal I/R and kidney injury
Intestinal I/R precipitates acute kidney injury (AKI) through combined hemodynamic and inflammatory mechanisms. There is a significant reduction in renal blood flow and tissue ATP levels following intestinal insult, impairing tubular function[68]. Histologically, the kidney displays changes akin to sepsis-induced AKI, including tubular dilation, epithelial flattening, and cast formation[69]. The gut-kidney axis plays a central role: intestinal barrier failure leads to the translocation of bacteria, toxins, and DAMPs, which directly damage renal tissue and incite local inflammation. Conversely, AKI-induced uremia and systemic inflammation further worsen intestinal integrity, creating a vicious cycle that fuels progressive renal dysfunction[70]. Currently, research predominantly focuses on the acute manifestations of AKI triggered by I/R injury, while its chronic progression remains poorly understood. This underscores the urgent need for further investigation into the long-term effects of intestinal I/R on renal health to develop effective preventive and management strategies.
Intestinal I/R and heart injury
Myocardial injury following intestinal I/R manifests as structural disarray, including myofiber derangement, interstitial edema, and nuclear pyknosis[71, 72]. The primary drivers are systemic inflammation and profound oxidative stress. The influx of inflammatory mediators and ROS during reperfusion disrupts cardiomyocyte electrophysiology and contractility, leading to arrhythmias and depressed function[73]. Oxidative stress also alters the expression of cardiac genes related to antioxidant defense[73]. Therapeutic strategies that enhance antioxidant capacity (e.g., hyperbaric oxygen[74]), modulate metabolism and inflammation (e.g., adenosine[75]), or reduce cardiac workload (e.g., atenolol[72]) have shown protective potential, highlighting the importance of targeting metabolic instability and oxidative damage.
Intestinal I/R and lung injury
The lung, with its extensive capillary network, is highly susceptible to I/R-induced damage, making acute lung injury/ARDS a common severe complication. The gut-lung axis is pivotal: microbial metabolites like succinate, whose balance is disrupted during I/R, can travel to the lungs, polarize alveolar macrophages via SUCNR1, and exacerbate injury[29, 76]. Moreover, bacterial translocation and systemic DAMPs (e.g., cold-inducible RNA-binding protein (CIRP) activate pulmonary TLR4, driving NF-κB and p38 MAPK pathways that intensify inflammation and endothelial damage[34, 77, 78].
Various interventions targeting these inflammatory pathways show therapeutic potential. The p38 MAPK inhibitor SB239063 alleviates I/R-induced lung injury by reducing IL-1β expression[43]. Similarly, curcumin and the sirtuin inhibitor FK866 attenuate lung inflammation and apoptosis via fine-tuning NF-κB signaling and reducing pro-inflammatory cytokine release[50, 53]. Beyond direct pathway inhibition, regulating related cellular functions is another important approach. For instance, I/R-associated oxidative stress is closely connected to mast cell degranulation. Agents such as propofol, sevoflurane, and resveratrol exert protective effects by stabilizing mast cells[79-81]. Ischemic preconditioning (IPO) and immediate ischemic postconditioning (IPC) during reperfusion also mitigate lung injury by suppressing peroxide generation, restoring superoxide dismutase (SOD) activity, and inhibiting inflammatory[82].
In addition to inflammation, oxidative stress and its mediated forms of cell death are key mechanisms of lung injury. Natural extracts, including Ginkgo biloba extract (EGb 761), artemisinin, and cilostazol, also exhibit lung-protective effects through antioxidant mechanisms[83-85]. Recently, the role of ferroptosis in ALI has garnered significant attention. Nrf2 can inhibit ferroptosis by upregulating the expression of molecules such as TERT and SLC7A11, thereby reducing alveolar epithelial cell apoptosis[86]. Furthermore, studies have revealed that the YAP/Nrf2 pathway alleviates acute lung injury by suppressing ferroptosis[87]. Additionally, celastrol has been shown to attenuate ferroptosis-mediated intestinal I/R-induced acute lung injury via Hippo-YAP signaling[88]. Inhibitor of apoptosis-stimulating protein of p53 and isoliquiritin also suppress ferroptosis by attenuating aberrant Fe2+ accumulation in lung tissue[89, 90]. Moreover, the PKCβII/p66Shc pathway is involved in regulating apoptosis, and its inhibition reduces caspase-3 cleavage[78, 91]. Notably, the endogenous antimicrobial peptide Beta-defensin-2 is upregulated following I/R injury, and moderate suppression of this upregulation may attenuate lung damage by inhibiting excessive inflammation and oxidative stress[92, 93], highlighting its complex role in injury pathogenesis. Recent studies have further expanded the intervention approaches. For instance, electroacupuncture has been shown to alleviate acute lung injury through the vagus-sympathetic nerve pathway[94], providing new evidence for lung protection strategies targeting neuroimmune regulation.
Collectively, lung injury following I/R involves the interplay of gut-lung axis dysfunction, activation of inflammatory signaling, oxidative stress, and multiple cell death pathways. Current research not only deepens the understanding of injury mechanisms but also provides a scientific basis for developing combined therapeutic strategies targeting inflammatory pathways, antioxidant defense, and the regulation of programmed cell death.
Intestinal I/R and brain injury
Intestinal I/R can induce remote brain injury characterized by neuroinflammation, blood-brain barrier disruption, and cognitive impairment. Mechanistically, this process is mediated by circulating factors, with enterogenic exosomes identified as key carriers of injurious signals that activate brain microglia. For instance, a recent study demonstrated that intestinal I/R injury influences hyaluronan homeostasis in the rat brain[95]. Microglial activation peaks around 24 hours post-reperfusion and is a central driver of neuronal oxidative stress and memory deficits[96]. The cerebrovascular barrier is compromised earlier, within hours of reperfusion[97]. Hall et al. previously reported that a rat model subjected to one hour of intestinal ischemia followed by two hours of reperfusion did not exhibit significant damage in the central nervous system[98]. This lack of damage may be attributed to the short duration of reperfusion, which likely did not reach the critical threshold necessary for inducing brain injury. Subsequent research has highlighted that enterogenic exosomes serve as key mediators of I/R-induced brain injury, contributing to memory decline through microglial activation[99]. Melatonin and small extracellular vesicles derived from Akkermansia muciniphila can attenuate microglial activation by modulating TLR pathways, thereby alleviating neuroinflammation and cognitive impairment[57, 100]. Additionally, the locus coeruleus norepinephrine (LCNE) system plays a significant role in mitigating cerebral injury. Dexmedetomidine preconditioning acts by activating central α2-adrenergic receptors, which inhibits the LCNE system, reduces brain norepinephrine levels, and subsequently stimulates cholinergic anti-inflammatory pathways to improve cognitive function[98].
The diagnosis and biomarkers of intestinal injury
Acute mesenteric ischemia (AMI), a critical manifestation of intestinal I/R injury, results from the abrupt interruption of mesenteric blood flow[1]. Despite its rarity, AMI carries a mortality rate exceeding 60%, largely attributable to diagnostic delays stemming from its nonspecific and often atypical clinical presentation. The classic symptom of severe, poorly localized abdominal pain is less common than more insidious "chronic-acute" forms[101, 102]. Therefore, heightened clinical suspicion, coupled with the judicious use of modern imaging and biomarkers, is paramount for timely diagnosis and improved outcomes.
Imaging modalities
Imaging is the cornerstone for confirming the diagnosis and assessing the extent of intestinal I/R injury.
Computed Tomography Angiography (CTA): CTA, particularly with multi-detector CT (MDCT) technology, is the first-line imaging modality for suspected AMI. It offers exceptional accuracy (sensitivity > 93%, specificity up to 100%) in detecting vascular occlusion, bowel wall abnormalities (e.g., edema, pneumatosis), and secondary signs of ischemia[103]. Its speed and widespread availability make it indispensable for urgent evaluation.
Magnetic Resonance Angiography (MRA): MRA serves as a valuable alternative, especially for patients with contraindications to iodinated contrast or for evaluating chronic mesenteric ischemia[104]. It provides excellent visualization of the proximal mesenteric vasculature. However, its utility may be limited in assessing distal branch occlusions or non-occlusive ischemia[105, 106]. Advanced techniques like 4D flow MRI are enhancing its diagnostic role[104, 107].
Doppler Ultrasound: While not a primary tool for diagnosing acute AMI due to operator dependency and technical limitations in an acute setting[108], Doppler ultrasound plays a significant role in the initial assessment and screening of chronic mesenteric ischemia. It effectively evaluates stenosis in the superior mesenteric and celiac arteries by measuring blood flow velocities[109-111].
Abdominal Radiography (X-ray): Plain abdominal X-rays have very limited diagnostic value in AMI. Findings are often non-specific and appear only in late stages (e.g., pneumoperitoneum from perforation), making it an unreliable tool for exclusion[108, 112]. Its use in this context is generally discouraged. In contrast, abdominal CT imaging is highly effective in detecting critical indicators such as pneumatosis intestinalis and portomesenteric venous gas, rendering it superior for this purpose. Therefore, abdominal CT should be prioritized for suspected AMI to enhance diagnostic accuracy and timeliness, ultimately optimizing patient care and treatment outcomes.
Circulating biomarkers
Biomarkers offer complementary, non-invasive tools to raise suspicion and support diagnosis, though none are pathognomonic alone.
D-dimer: D-dimer testing plays a crucial role in diagnosing AMI; however, its limitations in specificity and sensitivity necessitate the use of supplementary diagnostic methods[113, 114]. While a normal level may help rule out ischemia in low-risk patients, elevated levels (e.g., > 0.9 mg/L or > 1000 ng/ml) significantly increase the probability of AMI, especially when combined with risk factors like atrial fibrillation[115, 116]. It is best used as an adjunct to clinical assessment and imaging.
Intestinal fatty acid-binding protein (I-FABP): I-FABP, a cytosolic protein abundant in enterocytes, is released rapidly upon intestinal epithelial injury. It shows high sensitivity for detecting intestinal ischemia, particularly in conditions like strangulating bowel obstruction[117, 118]. Its detection in plasma or, notably, in urine provides a promising non-invasive method for early suspicion[117, 118], though standardized assays are needed for widespread clinical adoption.
Lactate: Serum or arterial lactate is a marker of tissue hypoperfusion and anaerobic metabolism. Elevated lactate is a concerning sign in suspected AMI, indicating advanced ischemia and potential bowel necrosis[119-122]. Although serum lactate measurements may lack absolute specificity for diagnosing AMI, they remain crucial for assessing the severity of the condition. Specifically, patients exhibiting symptoms of peritoneal inflammation and abdominal infections, along with lactate concentrations exceeding 2 mmol/L—especially when clinical suspicion focuses on peritonitis and small bowel compromise leading to necrosis—are strongly advised to undergo surgical exploration[123]. Nevertheless, it is imperative to exercise caution, as an isolated elevation in lactate levels, without supporting clinical evidence, is insufficient to differentiate between the onset of ischemia and irreparable intestinal damage.
Other Promising Biomarkers: Citrulline: A low plasma citrulline level reflects reduced functional enterocyte mass and shows promise as a marker of intestinal compromise[124, 125]. Procalcitonin (PCT): Serum PCT demonstrates predictive value for bowel ischemia and necrosis in cases of bowel obstruction[126-128]. Inflammatory Markers: The measurement of plasma IL-6 upon admission may serve as a predictive marker for intestinal ischemia and assist in the acute diagnosis of clinically suspicious cases[129-131]. Additionally, immature granulocyte count and delta neutrophil index are reliable indicators for evaluating intestinal necrosis in cases of mesenteric ischemia[132, 133]. Tissue-Specific Proteins: SM22 (released from damaged intestinal smooth muscle) and alpha-glutathione S-transferase are being explored as more specific indicators of bowel wall injury[134, 135].
The diagnosis of intestinal I/R injury requires a multimodal approach. A high index of clinical suspicion must trigger timely CTA imaging for definitive anatomical assessment. Biomarkers like D-dimer, I-FABP, and lactate serve as valuable adjuncts to raise early suspicion, triage patients, and gauge disease severity. The future lies in validating panels of biomarkers and integrating them with advanced imaging findings to enable earlier, more accurate diagnosis and risk stratification, ultimately improving the dire prognosis associated with this condition.
Preventive and therapeutic strategies and drugs for intestinal I/R injury
Advances in understanding the pathophysiology of intestinal I/R injury have catalyzed the shift from supportive care to mechanism-driven therapies. Current strategies aim to intercept the vicious cycle of oxidative stress, inflammatory cascades, and barrier failure. This section synthesizes these approaches into a coherent framework, spanning molecular interventions, regenerative medicine, microbiome modulation, and integrated clinical management.
Targeting core injury mechanisms
Antioxidant and Nrf2-activating strategies
Given that oxidative stress is a central executor of I/R damage, reinforcing endogenous antioxidant defenses has been a major therapeutic focus. The transcription factor Nrf2, as the master regulator of this response, represents a pivotal target. For instance, compounds such as isoquercitrin, dimethyl fumarate and bryostatin-1 confer protection by specifically activating the Nrf2/HO-1 pathway, thereby attenuating oxidative stress and secondary inflammation[136, 137]. Moreover, recent studies have revealed alternative mechanisms for mitigating oxidative damage. Coenzyme Q10 has been shown to alleviate intestinal I/R injury by inhibiting ferroptosis and lipid peroxidation[138], while obacunone suppresses mitochondrial ROS and MIF signaling to inhibit RIPK1/RIPK3/MLKL-mediated necroptosis in intestinal I/R injury[139]. Similarly, the mitochondria-targeted antioxidant MitoQ mitigates intestinal epithelial cell apoptosis, an effect closely associated with the activation of Nrf2 signaling and the stabilization of mitochondrial integrity[15]. Beyond direct Nrf2 activators, other agents operate through complementary pathways: Glutamine alleviates injury by inhibiting the xanthine oxidase/uric acid axis[140], while compounds like agmatine and naringin exert their effects by enhancing cellular glutathione reserves and reducing lipid peroxidation[141, 142]. Collectively, these findings underscore that boosting antioxidant capacity, particularly via the Nrf2 hub, is a validated strategy to mitigate the initial oxidative insult of I/R.
Modulation of non-coding RNAs
The regulatory landscape of I/R injury is profoundly influenced by non-coding RNAs (ncRNAs), especially microRNAs (miRNAs) and circular RNAs (circRNAs), which fine-tune gene expression in processes like apoptosis and inflammation. Dysregulation of specific ncRNAs is therefore mechanistically linked to injury progression. For example, the downregulation of miR-378 post-I/R permits enhanced caspase-3 activity and apoptosis, whereas its overexpression provides protection[143]. Likewise, the overexpression of miR-339-5p diminishes ROS production by targeting p66Shc, thereby mitigating I/R-induced damage[144]. In addition to modulating cell death and oxidative stress, miR-146a and miR-381-3p are crucial for dampening the immunoinflammatory response and promoting epithelial repair[145, 146]. Furthermore, the circular RNA CircEZH2_005, whose expression decreases after injury, has been shown to promote the proliferation of Lgr5+ stem cells, facilitating mucosal regeneration[147]. Thus, these ncRNAs not only serve as promising diagnostic biomarkers but also represent a novel class of therapeutic targets for RNA-based interventions.
Ischemic conditioning
IPO and IPC harness endogenous protective mechanisms, primarily by blunting the oxidative and inflammatory burst during early reperfusion. IPO exerts its benefit by reducing ROS generation and inhibiting the JAK/STAT signaling pathway, which in turn limits epithelial apoptosis[148, 149]. Complementarily, IPC appears to function through upregulation of aldose reductase and suppression of the TLR4-TRAF6 pathway[150, 151]. Critically, the protective window for these interventions is narrow; for instance, the synergy between IPO and IPC is most effective when applied during the initial moments of reperfusion[148]. This underscores the importance of timing in translating these powerful strategies into clinical practice.
Advanced and regenerative therapies
Stem cell and organoid-based therapy
Stem cell therapy, which involves the use of stem cells or their derivatives to repair damaged cellular structures, has gained widespread application in the treatment of various diseases, ranging from autoimmune disorders to organ ischemic injuries[152]. As a key component, mesenchymal stem cells (MSCs) have shown significant promise in addressing intestinal I/R injury, leveraging their self-renewal, multi-lineage differentiation, and potent immunomodulatory and paracrine functions[153-156]. It is noteworthy that although MSCs from different tissue sources (bone marrow, adipose tissue, and umbilical cord) share common core mechanisms like homing, immunomodulation, and paracrine signaling, their biological behaviors and therapeutic efficacy exhibit significant heterogeneity. This variability is primarily attributed to the distinct "tissue microenvironment imprint" carried by MSCs from each origin. For instance, bone marrow-derived MSCs can deliver miRNAs via extracellular vesicles to regulate the SREBF2/HMGB1 axis, thereby inhibiting ferroptosis[157]. Adipose-derived MSCs primarily exert their effects via the COX-2-PGE2 signaling axis[158], and by regulating macrophage polarization[159]; furthermore, combining them with melatonin results in a synergistic therapeutic effect[160]. Umbilical cord-derived MSCs demonstrate notable anti-apoptotic and reparative capacities[161]. Furthermore, hair follicle-derived MSCs alleviate intestinal I/R injury by modulating oxidative stress responses and reducing apoptosis[162]. Collectively, these findings illustrate the tissue-specific mechanisms of action of MSCs from various sources, providing a theoretical basis for selecting optimal cell origins in clinical applications. Advancements in organoid technology further complement stem cell-based approaches. For example, Zhang et al. demonstrated that intestinal organoid transplantation in mice enhances survival, promotes the self-renewal of intestinal stem cells, and improves the immune microenvironment following I/R injury. This effect is associated with the secretion of L-Malic acid (MA), which polarizes anti-inflammatory M2 macrophages and restores interleukin-10 levels through the action of SOCS2[36].
Microbiota-targeted interventions
The gastrointestinal microbiota and their metabolites have demonstrated significant therapeutic potential in the treatment of AMI. Probiotic-based strategies, such as the administration of Bifidobacterium bifidum PRL2010, Lactobacillus plantarum, and L. murinus, have been shown to mitigate intestinal I/R injury by preventing bacterial translocation and preserving barrier function[30, 163, 164]. Similarly, the probiotic VSL#3 has been shown to effectively reduce local tissue damage caused by elevated inflammatory mediators and the recruitment of immune cells[165]. Furthermore, Lactobacillus amplifies DHAMaR1 conversion to attenuate intestinal I/R injury via decreasing pyroptosis[166]. Beyond probiotics, microbiota-induced natural antibodies play a crucial role in the host's inflammatory response to both sterile and infectious damage. The gut symbiotic microbiota enhances intestinal immune vigilance by modulating the formation and recruitment of NETs[167].
Microbial metabolites also contribute significantly to intestinal protection. Short-chain fatty acids (SCFAs; e.g., butyrate, propionate, and acetate) help maintain mucosal integrity and attenuate neutrophil infiltration during I/R, highlighting their potential as preventive agents[168, 169]. The intestinal microbial metabolite pravastatin (PA) alleviates injury by enhancing interleukin-13 (IL-13) from innate lymphoid cells type 2 (ILC2)[41], while milnacipran promotes intestinal tolerance via the AHR/ILC3/IL-22 pathway[170]. Propionate mitigates damage by modulating mast cell function through the AhR/Notch1 axis[171], and microbiota-derived glutathione suppresses ferroptosis[172]. Additional metabolites such as capsiate (which activates TRPV1 to upregulate Gpx4 and inhibit ferroptosis)[21], petroselinic acid (which activates the AMPK-mTOR pathway to suppress apoptosis)[173], and Indole-3-lactic acid (which enhances epithelial stem cell function through YAP and Nrf2 activation and protects the gut-vascular barrier through AhR/Nrf2/STAT3-mediated claudin-2 downregulation)[174, 175]. These findings underscore the potential of the gut microbiome and its metabolic products in maintaining intestinal integrity and regulating immune responses, highlighting their significant impact on AMI treatment and suggesting new directions for future therapeutic strategies.
Clinical management principles
The effective management of acute mesenteric ischemia (AMI), the stark clinical manifestation of I/R, demands a rapid, sequential, and multidisciplinary approach. Management must begin with aggressive fluid resuscitation to restore perfusion, accompanied by correction of acidosis and electrolyte imbalances. Crucially, early empirical broad-spectrum antibiotics are mandatory to mitigate the imminent risk of bacterial translocation and sepsis[176-178]. Therefore, a comprehensive approach that includes fluid management, correction of electrolyte and acid-base imbalances, hemodynamic monitoring, and proactive antibiotic treatment serves as the foundation for minimizing complications and optimizing the physiological state.
In the treatment of AMI, timely surgical intervention is critical; it aims to restore blood supply to the affected intestinal segment, resect all non-viable bowel areas, and preserve as much healthy intestine as possible. Surgical treatment is vital for ensuring intestinal viability, as failure to identify and manage non-viable intestine can lead to multi-organ dysfunction and increased patient mortality[179]. Acute peritonitis may indicate bowel infarction, necessitating immediate laparoscopy or laparotomy. Although surgical intervention enhances survival rates, mortality remains high in cases of acute arterial thrombosis. Nonetheless, nearly 40% of patients who undergo endovascular revascularization survive for one month post-operation, suggesting that endovascular treatment is an effective and cost-efficient management strategy for AMI[180]. Percutaneous stent placement and treatment of SMA occlusion yield significant benefits. Puncture aspiration and thrombolysis enhance outcomes in selected patient groups[181]. Subsequent laparoscopic examinations have minimal impact on outcomes[182]. While surgical treatment remains crucial, emerging endovascular strategies provide alternative options tailored to individual patient conditions. After successful intervention, long-term anticoagulation/antiplatelet therapy is required to prevent recurrence[183, 184]. Furthermore, lifelong monitoring with imaging surveillance is recommended to detect and manage late complications such as restenosis[180].
The therapeutic arsenal for intestinal I/R injury is expanding from generic support to mechanism-targeted strategies, as synthesized in Table 1. From a translational perspective, several pathways and biomarkers emerging from experimental and early clinical studies show particular promise for near-term clinical application. Circulating markers of intestinal epithelial injury, including I-FABP, D-lactate, and citrulline, have been proposed as early indicators of intestinal barrier disruption. In parallel, inflammatory mediators such as HMGB1, pro-inflammatory cytokines, and markers of NET formation reflect systemic immune activation and correlate with distant organ injury. At the mechanistic level, signaling pathways involving TLR/MyD88 activation, oxidative stress responses, the Nrf2/HO-1 antioxidant pathway, and microbiota-derived metabolic signaling represent attractive therapeutic targets. Clinically feasible strategies to attenuate intestinal I/R-induced systemic injury may thus include interventions aimed at preserving microcirculatory integrity, modulating innate immune activation, or restoring gut microbiota homeostasis. Looking forward, the translational challenge lies in advancing ncRNA therapeutics and stem cell applications from bench to bedside, personalizing microbiota-targeted interventions, and, most importantly, integrating these novel adjuvant approaches into the time-sensitive clinical management framework to improve the prognosis of this condition.
Summary of Mechanism-Targeted Interventions and Strategies for Intestinal I/R Injury
| Category | Intervention / Biomarker | Model & Timing / Dose | Mechanism / Key Effects | Reference |
|---|---|---|---|---|
| Intestinal I/R Strategies | Ischemic preconditioning (IPC) | SMA occlusion 5 min + reperfusion 5 min × 3 | ↓ ROS, ↑ SOD, ↓ inflammatory cytokines, ↓ apoptosis (caspase-3/Bax ↓, Bcl-2 ↑) | Wang, Zhidong et al. |
| Ischemic postconditioning (IPoC) | Immediate 30 s reperfusion/30 s occlusion × 3 | Similar to IPC; ineffective if delayed by 3 min | Liu, Ke-Xuan et al. | |
| Osthole | i.p. 10/50 mg/kg, 30 min before ischemia | Anti-inflammatory and antioxidant: ↓ ROS, ↑ SOD, ↓ neutrophil infiltration | Mo, Li-Qun et al. | |
| EGb 761 (Ginkgo extract) | p.o. 100 mg/kg for 7 days pre-modeling | Antioxidant, ↓ neutrophils and iNOS-NO | Liu, Ke-Xuan et al. | |
| Curcumin | i.v. 1/5 mg/kg, 50 min ischemia | NF-κB inhibition, ↓ IL-6, ICAM-1, neutrophil infiltration | Fan, Zhe et al. | |
| Fenofibrate | i.p. 100 mg/kg, 1 h before ischemia | PPARα agonist; inhibits NF-κB p65, reduces inflammation & apoptosis | Zhu, Qiankun et al. | |
| MSC-derived exosomes | i.v. 500 μL (3×10^6 MSCs) at reperfusion | TLR4/NF-κB suppression, ↓ inflammation | Liu, Jianpei et al. | |
| Distant Organ Protection | Reba | p.o. 100 mg/kg/day | ↑SIRT1, ↑β-catenin, ↑ FOXO1, ↓NF-κB p65 | Elwany, Nisreen E et al. |
| atenolol | intravenous atenolol infusion (2 mg/kg) | ↑malonaldehyde (p=0.001), ↓ TNF-α (p=0.001) | Okada, Mieko et al. | |
| SB239063 (p38MAPK inhibitor) | i.p. 10 mg/kg, 1 h post-op | ↓ p38MAPK → ↓ IL-1β / AQP4, ↓ lung injury | Zheng, De-Yi et al. | |
| FK866 (visfatin inhibitor) | i.p. 10 mg/kg at reperfusion | ↓ Visfatin → NF-κB suppression, ↓ inflammation & apoptosis | Matsuda, Akihisa et al. | |
| Melatonin → Brain | i.p. 75/150 mg/kg at reperfusion | TLR4/MyD88 inhibition in microglia, ↓ neuroinflammation & apoptosis | Yang, Bo et al. | |
| Diagnostics / Imaging | CT / CTA | MDCT / Biphasic MDCT Angiography | AMI diagnosis: Sensitivity 93-96%, Specificity 96-100%; non-enhancement predicts necrosis | Menke, Jan. |
| D-dimer | Serum test | Sensitivity 96%, specificity 18-40%; > 1.5 mg/L suggests SMA occlusion risk | Chiu, Yu-Hui et al. | |
| I-FABP | Plasma / urine test | Plasma: Sens 79%, Spec 91%; Urine AUC 0.88; > 3.1 ng/mL for small intestine ischemia | Kanda, T et al. | |
| Myoglobin, AST, PCT, PALM score | Combined scoring system | AUROC 0.93; PALM ≥14 predicts high risk | Zogheib, Elie et al. | |
| Clinical Treatments | Crystalloid resuscitation | Up to 100 mL/kg or 32 mL/kg/h | Stabilizes hemodynamics, corrects acidosis/hyperkalemia | Lagoa, Claudio Esteves et al. |
| Oral antibiotics | Gentamicin 80 mg/d + Metronidazole 1.5 g/d | ↓ ITIN risk (HR 0.16) | Nuzzo, Alexandre et al. | |
| Surgery / endovascular revascularization | Open vs PTA + stenting | Revascularization reduces 30-day mortality (42% vs 62%) | Erben, Young et al. | |
| Papaverine ± PGE1 | Intra-arterial SMA infusion | ↑ SMA perfusion; improved survival in NOMI (risk diff -11.6%) | Boley, S J et al. | |
| Teduglutide (GLP-2 analog) | s.c. 0.05 mg/kg/day | ↓ TPN by 4.4 L/week; ↑ mucosal mass (citrulline) | Jeppesen, Palle B et al. | |
| IVIG | i.v. 0.5-1 g/kg | Clears C3b/C5a, inhibits complement deposition, protects local & distant organs | Anderson, Jimie et al. | |
| Probiotics / SCFA (e.g., B. bifidum, VSL#3, butyrate) | Multiple strains and metabolites | ↓ BT, inflammation, apoptosis, NETs; ↑ barrier, IL-10/IgG; ↓ TLR4/TRIF | Qiao, Yingli et al. |
Others
Identifying early and specific biomarkers for gastrointestinal ischemic injury is essential[177]. Future research should concentrate on studying survivors of gastrointestinal ischemic injury to identify predictors of bowel resection and short bowel syndrome, as well as to develop visceral-mesenteric vasoactive agents for ischemia-related vasospasm. Currently, no single biomarker meets the gold standard diagnostic criteria of high sensitivity and specificity. Therefore, the comprehensive application of multiple laboratory indicators holds significant clinical value for assessing the likelihood of acute mesenteric ischemia. Future studies should continue to explore and validate novel biomarkers to improve diagnostic efficacy, and combining morphological techniques (such as microscopic imaging and tissue viability assessment) can further enhance diagnostic accuracy[185]. Establish 'intestinal stroke centers' that emphasize multidisciplinary collaboration, focusing on bowel resection, revascularization, and intensive care to prevent progression to multi-organ failure[179]. Despite the limited diagnostic capabilities of many current biomarkers, future research should aim to enhance diagnostic accuracy and efficiency through the integration of technologies such as artificial intelligence and machine learning. Strengthening the translation of basic research into clinical applications will facilitate the adoption of new therapies and technologies.
In conclusion, I/R injury represents a lethal, self-amplifying cascade in which microcirculatory failure and barrier disruption transform a localized ischemic event into a systemic inflammatory disorder. Acting as a central amplifier of immune and metabolic stress, the injured intestine promotes the dissemination of gut-derived danger signals, inflammatory mediators, and toxic metabolites, thereby driving distant organ injury and multiple organ dysfunction. This review systematically integrates the core mechanisms underlying intestinal I/R injury—including oxidative stress, regulated cell death, gut dysbiosis, and dysregulated immune signaling—within a unified pathophysiological framework linking local intestinal damage to systemic consequences. Based on these mechanistic insights, we summarize emerging diagnostic approaches and therapeutic strategies, such as ischemic conditioning, stem cell therapy, and microbiota-targeted interventions, that aim to interrupt key nodes of this injury cascade. Although timely revascularization remains the cornerstone of clinical management, precision interventions targeting specific components of the gut-centered inflammatory axis hold increasing translational promise. Future efforts should focus on translating these mechanistic advances into clinically validated tools through multi-omics-based risk stratification and personalized therapeutic strategies, with the ultimate goal of reducing the persistently high mortality associated with intestinal I/R injury.
Acknowledgements
Figures were created using BioRender (www.biorender.com). During the preparation of this manuscript, the authors used ChatGPT-4o for language polishing and grammar correction. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the final content of the publication. No AI tools were used as authors of this paper.
Funding
This work was funded by the National Natural Science Foundation of China (82372169 to F.D; 82472185 to J.-J.H), the Key Program of National Natural Science Foundation, Beijing, China (82330067 to Kexuan Liu), the China International Medical Exchange Foundation (Grant No: [Z-2017-24-2421] to F.D.), the Guangzhou Basic and Applied Basic Research Project Special Fund (Young Doctoral "Qihang" Program) (Grant No: [2025A04J4103] to F.D.), the Southern Medical University Distinguished Recruit Talent Research Startup Fund (Grant No: [R10101063] to F.D.), the Southern Hospital President's Fund (Grant No: [K50112078] to F.D.).
Data availability statements
The authors confirm that the data supporting the findings of this study are available within the article.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49:1359-77
2. Stamatakos M, Stefanaki C, Mastrokalos D, Arampatzi H, Safioleas P, Chatziconstantinou C. et al. Mesenteric ischemia: still a deadly puzzle for the medical community. Tohoku J Exp Med. 2008;216:197-204
3. Acosta S. Epidemiology of mesenteric vascular disease: clinical implications. Semin Vasc Surg. 2010;23:4-8
4. Smalcova J, Suen J, Huptych M, Franek O, Kavalkova P, Brodska HL. et al. The significance of possible non-occlusive mesenteric ischemia in relation to neurological outcomes in patients with refractory cardiac arrest - Secondary analysis of the Prague OHCA study. Resuscitation. 2025;214:110642
5. Hundscheid IHR, Schellekens D, Grootjans J, Derikx JPM, Buurman WA, Dejong CHC. et al. Females Are More Resistant to Ischemia-Reperfusion-induced Intestinal Injury Than Males: A Human Study. Ann Surg. 2020;272:1070-9
6. Vollmar B, Menger MD. Intestinal ischemia/reperfusion: microcirculatory pathology and functional consequences. Langenbecks Arch Surg. 2011;396:13-29
7. Deitch EA. Role of the gut lymphatic system in multiple organ failure. Curr Opin Crit Care. 2001;7:92-8
8. Tassopoulos A, Chalkias A, Papalois A, Iacovidou N, Xanthos T. The effect of antioxidant supplementation on bacterial translocation after intestinal ischemia and reperfusion. Redox Rep. 2017;22:1-9
9. Leaphart CL, Tepas JJ 3rd. The gut is a motor of organ system dysfunction. Surgery. 2007;141:563-9
10. Chopyk DM, Grakoui A. Contribution of the Intestinal Microbiome and Gut Barrier to Hepatic Disorders. Gastroenterology. 2020;159:849-63
11. Verdu EF, Galipeau HJ, Jabri B. Novel players in coeliac disease pathogenesis: role of the gut microbiota. Nat Rev Gastroenterol Hepatol. 2015;12:497-506
12. Albillos A, de Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol. 2020;72:558-77
13. Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev. 2014;94:329-54
14. Brand MD, Goncalves RL, Orr AL, Vargas L, Gerencser AA, Borch Jensen M. et al. Suppressors of Superoxide-H(2)O(2) Production at Site I(Q) of Mitochondrial Complex I Protect against Stem Cell Hyperplasia and Ischemia-Reperfusion Injury. Cell Metab. 2016;24:582-92
15. Hu Q, Ren J, Li G, Wu J, Wu X, Wang G. et al. The mitochondrially targeted antioxidant MitoQ protects the intestinal barrier by ameliorating mitochondrial DNA damage via the Nrf2/ARE signaling pathway. Cell Death Dis. 2018;9:403
16. Pabst O, Hornef MW, Schaap FG, Cerovic V, Clavel T, Bruns T. Gut-liver axis: barriers and functional circuits. Nat Rev Gastroenterol Hepatol. 2023;20:447-61
17. Zhang X, Wu J, Liu Q, Li X, Li S, Chen J. et al. mtDNA-STING pathway promotes necroptosis-dependent enterocyte injury in intestinal ischemia reperfusion. Cell Death Dis. 2020;11:1050
18. Ikeda H, Suzuki Y, Suzuki M, Koike M, Tamura J, Tong J. et al. Apoptosis is a major mode of cell death caused by ischaemia and ischaemia/reperfusion injury to the rat intestinal epithelium. Gut. 1998;42:530-7
19. Jia Y, Cui R, Wang C, Feng Y, Li Z, Tong Y. et al. Metformin protects against intestinal ischemia-reperfusion injury and cell pyroptosis via TXNIP-NLRP3-GSDMD pathway. Redox Biol. 2020;32:101534
20. Li Y, Feng D, Wang Z, Zhao Y, Sun R, Tian D. et al. Ischemia-induced ACSL4 activation contributes to ferroptosis-mediated tissue injury in intestinal ischemia/reperfusion. Cell Death Differ. 2019;26:2284-99
21. Deng F, Zhao BC, Yang X, Lin ZB, Sun QS, Wang YF. et al. The gut microbiota metabolite capsiate promotes Gpx4 expression by activating TRPV1 to inhibit intestinal ischemia reperfusion-induced ferroptosis. Gut Microbes. 2021;13:1-21
22. Li S, Zhou Y, Gu X, Zhang X, Jia Z. NLRX1/FUNDC1/NIPSNAP1-2 axis regulates mitophagy and alleviates intestinal ischaemia/reperfusion injury. Cell Prolif. 2021;54:e12986
23. Li R, Wang A, Cao J, Li K, Zhang H, Wang Y. et al. Single-cell RNA sequencing reveals the role of neutrophils in intestinal ischemia-reperfusion injury in mice. Life Sci. 2026;388:124208
24. Qin X, Sheth SU, Sharpe SM, Dong W, Lu Q, Xu D. et al. The mucus layer is critical in protecting against ischemia-reperfusion-mediated gut injury and in the restitution of gut barrier function. Shock. 2011;35:275-81
25. Iacob S, Iacob DG. Infectious Threats, the Intestinal Barrier, and Its Trojan Horse: Dysbiosis. Front Microbiol. 2019;10:1676
26. Kip AM, Ceulemans LJ, Hundscheid IHR, Canovai E, Hartog H, Brown RM. et al. Paneth Cell Alterations During Ischemia-reperfusion, Follow-up, and Graft Rejection After Intestinal Transplantation. Transplantation. 2020;104:1952-8
27. Deng F, Lin ZB, Sun QS, Min Y, Zhang Y, Chen Y. et al. The role of intestinal microbiota and its metabolites in intestinal and extraintestinal organ injury induced by intestinal ischemia reperfusion injury. Int J Biol Sci. 2022;18:3981-92
28. Chen J, Wang Y, Shi Y, Liu Y, Wu C, Luo Y. Association of Gut Microbiota with Intestinal Ischemia/Reperfusion Injury. Front Cell Infect Microbiol. 2022;12:962782
29. Wang YH, Yan ZZ, Luo SD, Hu JJ, Wu M, Zhao J. et al. Gut microbiota-derived succinate aggravates acute lung injury after intestinal ischaemia/reperfusion in mice. Eur Respir J. 2023 61
30. Hu J, Deng F, Zhao B, Lin Z, Sun Q, Yang X. et al. Lactobacillus murinus alleviate intestinal ischemia/reperfusion injury through promoting the release of interleukin-10 from M2 macrophages via Toll-like receptor 2 signaling. Microbiome. 2022;10:38
31. Li X, Hou M, Lyu J, Min X, Han X, Wang X. et al. Gut microbiota-derived arginine metabolism mitigates intestinal ischemia-reperfusion injury. J Transl Med. 2025;23:1215
32. Meyer F, Wendling D, Demougeot C, Prati C, Verhoeven F. Cytokines and intestinal epithelial permeability: A systematic review. Autoimmun Rev. 2023;22:103331
33. Honda M, Kadohisa M, Yoshii D, Komohara Y, Hibi T. Intravital imaging of immune responses in intestinal inflammation. Inflamm Regen. 2023;43:9
34. Victoni T, Coelho FR, Soares AL, de Freitas A, Secher T, Guabiraba R. et al. Local and remote tissue injury upon intestinal ischemia and reperfusion depends on the TLR/MyD88 signaling pathway. Med Microbiol Immunol. 2010;199:35-42
35. Zhan Y, Ling Y, Deng Q, Qiu Y, Shen J, Lai H. et al. HMGB1-Mediated Neutrophil Extracellular Trap Formation Exacerbates Intestinal Ischemia/Reperfusion-Induced Acute Lung Injury. J Immunol. 2022;208:968-78
36. Zhang FL, Hu Z, Wang YF, Zhang WJ, Zhou BW, Sun QS. et al. Organoids transplantation attenuates intestinal ischemia/reperfusion injury in mice through L-Malic acid-mediated M2 macrophage polarization. Nat Commun. 2023;14:6779
37. Benakis C, Brea D, Caballero S, Faraco G, Moore J, Murphy M. et al. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nat Med. 2016;22:516-23
38. Chu C, Wang X, Yang C, Chen F, Shi L, Xu W. et al. Neutrophil extracellular traps drive intestinal microvascular endothelial ferroptosis by impairing Fundc1-dependent mitophagy. Redox Biol. 2023;67:102906
39. Chu C, Wang X, Chen F, Yang C, Shi L, Xu W. et al. Neutrophil extracellular traps aggravate intestinal epithelial necroptosis in ischaemia-reperfusion by regulating TLR4/RIPK3/FUNDC1-required mitophagy. Cell Prolif. 2024;57:e13538
40. Kang J, Loh K, Belyayev L, Cha P, Sadat M, Khan K. et al. Type 3 innate lymphoid cells are associated with a successful intestinal transplant. Am J Transplant. 2021;21:787-97
41. Deng F, Hu JJ, Yang X, Sun QS, Lin ZB, Zhao BC. et al. Gut Microbial Metabolite Pravastatin Attenuates Intestinal Ischemia/Reperfusion Injury Through Promoting IL-13 Release from Type II Innate Lymphoid Cells via IL-33/ST2 Signaling. Front Immunol. 2021;12:704836
42. Zheng SY, Fu XB, Xu JG, Zhao JY, Sun TZ, Chen W. Inhibition of p38 mitogen-activated protein kinase may decrease intestinal epithelial cell apoptosis and improve intestinal epithelial barrier function after ischemia- reperfusion injury. World J Gastroenterol. 2005;11:656-60
43. Zheng DY, Zhou M, Jin J, He M, Wang Y, Du J. et al. Inhibition of P38 MAPK Downregulates the Expression of IL-1β to Protect Lung from Acute Injury in Intestinal Ischemia Reperfusion Rats. Mediators Inflamm. 2016;2016:9348037
44. Zhang Y, Jing Y, Liao H, Ding K, Zhang L, Liao S. et al. STING deficiency alleviates ischemia-reperfusion injury via JAK2/STAT3-mediated macrophage polarization and autophagy. Int Immunopharmacol. 2026;168:115784
45. Xiong LL, Tan Y, Ma HY, Dai P, Qin YX, Yang RA. et al. Administration of SB239063, a potent p38 MAPK inhibitor, alleviates acute lung injury induced by intestinal ischemia reperfusion in rats associated with AQP4 downregulation. Int Immunopharmacol. 2016;38:54-60
46. Li L, Shu F, Wang XQ, Wang F, Cai L, Zhao X. et al. Propofol alleviates intestinal ischemia/reperfusion injury in rats through p38 MAPK/NF-κB signaling pathway. Eur Rev Med Pharmacol Sci. 2021;25:1574-81
47. Liu XM, Chen QH, Hu Q, Liu Z, Wu Q, Liang SS. et al. Dexmedetomidine protects intestinal ischemia-reperfusion injury via inhibiting p38 MAPK cascades. Exp Mol Pathol. 2020;115:104444
48. Liu Z, Hu Q, Chen Q, Wu Q, Liang S, Nie J. et al. Polypeptide Nanomicrospheres Delivering Dexmedetomidine Mitigate Intestinal Ischemia/Reperfusion Injury by Regulating Autophagy and Inflammation. Antioxid Redox Signal. 2025
49. Chen LW, Egan L, Li ZW, Greten FR, Kagnoff MF, Karin M. The two faces of IKK and NF-kappaB inhibition: prevention of systemic inflammation but increased local injury following intestinal ischemia-reperfusion. Nat Med. 2003;9:575-81
50. Fan Z, Yao J, Li Y, Hu X, Shao H, Tian X. Anti-inflammatory and antioxidant effects of curcumin on acute lung injury in a rodent model of intestinal ischemia reperfusion by inhibiting the pathway of NF-Kb. Int J Clin Exp Pathol. 2015;8:3451-9
51. Liu J, Li B, Liu J, Qiu F, Diao Y, Lei Y. et al. Phenolic Acids from Fructus Chebulae Immaturus Alleviate Intestinal Ischemia-Reperfusion Injury in Mice through the PPARα/NF-κB Pathway. Molecules. 2022 27
52. Almoiliqy M, Wen J, Xu B, Sun YC, Lian MQ, Li YL. et al. Cinnamaldehyde protects against rat intestinal ischemia/reperfusion injuries by synergistic inhibition of NF-κB and p53. Acta Pharmacol Sin. 2020;41:1208-22
53. Matsuda A, Yang WL, Jacob A, Aziz M, Matsuo S, Matsutani T. et al. FK866, a visfatin inhibitor, protects against acute lung injury after intestinal ischemia-reperfusion in mice via NF-κB pathway. Ann Surg. 2014;259:1007-17
54. Abdel-Hamid HA, Ali DME, Hakim SBA, Ibrahim MFG. Unraveling the protective mechanisms of Angiotensin-(1-7) in Intestinal Ischemia-Reperfusion injury: Interplay between Nrf-2/HO-1 and NF-κB/NLRP3 pathways. Life Sci. 2025;379:123898
55. Xu H, He TQ, Chen SY, Shi RR, Xu J, Xing YR. et al. Isoquercitrin mitigates intestinal ischemia-reperfusion injury by regulating intestinal flora and inhibiting NLRP3 inflammasome activation. Redox Biol. 2025;86:103803
56. Chai D, Zhang L, Xi S, Cheng Y, Jiang H, Hu R. Nrf2 Activation Induced by Sirt1 Ameliorates Acute Lung Injury After Intestinal Ischemia/Reperfusion Through NOX4-Mediated Gene Regulation. Cell Physiol Biochem. 2018;46:781-92
57. Yang B, Zhang LY, Chen Y, Bai YP, Jia J, Feng JG. et al. Melatonin alleviates intestinal injury, neuroinflammation and cognitive dysfunction caused by intestinal ischemia/reperfusion. Int Immunopharmacol. 2020;85:106596
58. Huang QQ, Koessler RE, Birkett R, Perlman H, Xing L, Pope RM. TLR2 deletion promotes arthritis through reduction of IL-10. J Leukoc Biol. 2013;93:751-9
59. Turnage RH, Kadesky KM, Myers SI, Guice KS, Oldham KT. Hepatic hypoperfusion after intestinal reperfusion. Surgery. 1996;119:151-60
60. Uchiyama T, Delude RL, Fink MP. Dose-dependent effects of ethyl pyruvate in mice subjected to mesenteric ischemia and reperfusion. Intensive Care Med. 2003;29:2050-8
61. Wen S, Li X, Ling Y, Chen S, Deng Q, Yang L. et al. HMGB1-associated necroptosis and Kupffer cells M1 polarization underlies remote liver injury induced by intestinal ischemia/reperfusion in rats. Faseb j. 2020;34:4384-402
62. Horie Y, Wolf R, Miyasaka M, Anderson DC, Granger DN. Leukocyte adhesion and hepatic microvascular responses to intestinal ischemia/reperfusion in rats. Gastroenterology. 1996;111:666-73
63. Hayase N, Doi K, Hiruma T, Matsuura R, Hamasaki Y, Noiri E. et al. Recombinant Thrombomodulin on Neutrophil Extracellular Traps in Murine Intestinal Ischemia-Reperfusion. Anesthesiology. 2019;131:866-82
64. Zhao J, Chen XD, Yan ZZ, Huang WF, Liu KX, Li C. Gut-Derived Exosomes Induce Liver Injury After Intestinal Ischemia/Reperfusion by Promoting Hepatic Macrophage Polarization. Inflammation. 2022;45:2325-38
65. Wang J, Qiao L, Li Y, Yang G. Ginsenoside Rb1 attenuates intestinal ischemia-reperfusion- induced liver injury by inhibiting NF-kappaB activation. Exp Mol Med. 2008;40:686-98
66. Hartmann R, Licks F, Schemitt EG, Colares JR, Da Silva J, Moura RM. et al. Effect of glutamine on liver injuries induced by intestinal ischemia-reperfusion in rats. Nutr Hosp. 2017;34:548-54
67. Jing HR, Luo FW, Liu XM, Tian XF, Zhou Y. Fish oil alleviates liver injury induced by intestinal ischemia/reperfusion via AMPK/SIRT-1/autophagy pathway. World J Gastroenterol. 2018;24:833-43
68. LaNoue JL Jr, Turnage RH, Kadesky KM, Guice KS, Oldham KT, Myers SI. The effect of intestinal reperfusion on renal function and perfusion. J Surg Res. 1996;64:19-25
69. Lai HJ, Zhan YQ, Qiu YX, Ling YH, Zhang XY, Chang ZN. et al. HMGB1 signaling-regulated endoplasmic reticulum stress mediates intestinal ischemia/reperfusion-induced acute renal damage. Surgery. 2021;170:239-48
70. Chen R, Zeng Z, Zhang YY, Cao C, Liu HM, Li W. et al. Ischemic postconditioning attenuates acute kidney injury following intestinal ischemia-reperfusion through Nrf2-regulated autophagy, anti-oxidation, and anti-inflammation in mice. Faseb j. 2020;34:8887-901
71. Zickri MB, Embaby A, Metwally HG. Experimental study on the effect of intravenous stem cell therapy on intestinal ischemia reperfusion induced myocardial injury. Int J Stem Cells. 2013;6:121-8
72. Okada M, Falcão LFR, Ferez D, Martins JL, Errante PR, Rodrigues FSM. et al. Effect of atenolol pre-treatment in heart damage in a model of intestinal ischemia-reperfusion. Acta Cir Bras. 2017;32:964-72
73. Somaio Neto F, Ikejiri AT, Bertoletto PR, Chaves JC, Teruya R, Fagundes DJ. et al. Gene expression related to oxidative stress in the heart of mice after intestinal ischemia. Arq Bras Cardiol. 2014;102:165-73
74. Somaio FN, Ikejiri AT, Bertoletto PR, Chaves JC, Teruya R, Fagundes DJ. Hyperbaric oxygenation and the genic expression related to oxidative stress in the heart of mice during intestinal ischemia and reperfusion. Acta Cir Bras. 2017;32:913-23
75. Montero MF, Saurim R, Bonservizi WG, Koike MK, Taha MO. Heart injury following intestinal ischemia reperfusion in rats is attenuated by association of ischemic preconditioning and adenosine. Acta Cir Bras. 2014;29(Suppl 2):67-71
76. Wypych TP, Wickramasinghe LC, Marsland BJ. The influence of the microbiome on respiratory health. Nat Immunol. 2019;20:1279-90
77. Ben DF, Yu XY, Ji GY, Zheng DY, Lv KY, Ma B. et al. TLR4 mediates lung injury and inflammation in intestinal ischemia-reperfusion. J Surg Res. 2012;174:326-33
78. Cen C, McGinn J, Aziz M, Yang WL, Cagliani J, Nicastro JM. et al. Deficiency in cold-inducible RNA-binding protein attenuates acute respiratory distress syndrome induced by intestinal ischemia-reperfusion. Surgery. 2017;162:917-27
79. Zhao W, Zhou S, Yao W, Gan X, Su G, Yuan D. et al. Propofol prevents lung injury after intestinal ischemia-reperfusion by inhibiting the interaction between mast cell activation and oxidative stress. Life Sci. 2014;108:80-7
80. Huang X, Zhao W, Hu D, Han X, Wang H, Yang J. et al. Resveratrol efficiently improves pulmonary function via stabilizing mast cells in a rat intestinal injury model. Life Sci. 2017;185:30-7
81. Luo C, Yuan D, Zhao W, Chen H, Luo G, Su G. et al. Sevoflurane ameliorates intestinal ischemia-reperfusion-induced lung injury by inhibiting the synergistic action between mast cell activation and oxidative stress. Mol Med Rep. 2015;12:1082-90
82. Liu KX, Li YS, Huang WQ, Li C, Liu JX, Li Y. Immediate but not delayed postconditioning during reperfusion attenuates acute lung injury induced by intestinal ischemia/reperfusion in rats: comparison with ischemic preconditioning. J Surg Res. 2009;157:e55-62
83. Liu KX, Wu WK, He W, Liu CL. Ginkgo biloba extract (EGb 761) attenuates lung injury induced by intestinal ischemia/reperfusion in rats: roles of oxidative stress and nitric oxide. World J Gastroenterol. 2007;13:299-305
84. Ji T, Chen M, Liu Y, Jiang H, Li N, He X. Artesunate alleviates intestinal ischemia/reperfusion induced acute lung injury via up-regulating AKT and HO-1 signal pathway in mice. Int Immunopharmacol. 2023;122:110571
85. Gendy AM, Amin MM, Al-Mokaddem AK, Abd Ellah MF. Cilostazol mitigates mesenteric ischemia/reperfusion-induced lung lesion: Contribution of PPAR-γ, NF-κB, and STAT3 crosstalk. Life Sci. 2021;266:118882
86. Dong H, Xia Y, Jin S, Xue C, Wang Y, Hu R. et al. Nrf2 attenuates ferroptosis-mediated IIR-ALI by modulating TERT and SLC7A11. Cell Death Dis. 2021;12:1027
87. Tang L, Yang C, Peng Y, Liu M, Wei N, Fan X. et al. YAP/Nrf2 suppresses ferroptosis to alleviate acute lung injury induced by intestinal ischemia/reperfusion. Redox Biol. 2025;86:103811
88. Fan X, Ai Y, Mu G, Wang F, Feng J, Jia J. et al. Celastrol attenuates ferroptosis-mediated intestinal ischemia/reperfusion-induced acute lung injury via Hippo-YAP signaling. Phytomedicine. 2025;143:156865
89. Li Y, Cao Y, Xiao J, Shang J, Tan Q, Ping F. et al. Inhibitor of apoptosis-stimulating protein of p53 inhibits ferroptosis and alleviates intestinal ischemia/reperfusion-induced acute lung injury. Cell Death Differ. 2020;27:2635-50
90. Zhongyin Z, Wei W, Juan X, Guohua F. Isoliquiritin apioside relieves intestinal ischemia/reperfusion-induced acute lung injury by blocking Hif-1α-mediated ferroptosis. Int Immunopharmacol. 2022;108:108852
91. Wang G, Chen Z, Zhang F, Jing H, Xu W, Ning S. et al. Blockade of PKCβ protects against remote organ injury induced by intestinal ischemia and reperfusion via a p66shc-mediated mitochondrial apoptotic pathway. Apoptosis. 2014;19:1342-53
92. Li Y, Wu XB, Li JG, Lin YJ, Chen HL, Song H. et al. Enteral supplementation of alanyl-glutamine attenuates the up-regulation of beta-defensin-2 protein in lung injury induced by intestinal ischemia reperfusion in rats. Int J Surg. 2014;12:1181-6
93. Liu KX, Chen SQ, Zhang H, Guo JY, Li YS, Huang WQ. Intestinal ischaemia/reperfusion upregulates beta-defensin-2 expression and causes acute lung injury in the rat. Injury. 2009;40:950-5
94. Lv S, Ma C, Fu W, Hu C, Han X, Chen J. et al. Electroacupuncture alleviates intestinal ischemia-reperfusion-induced acute lung injury via the vagus-sympathetic nerve pathway. Int Immunopharmacol. 2025;165:115484
95. Bosi A, Baranzini N, Ponti A, Moretto P, Moro E, Crema F. et al. Intestinal Ischemia/Reperfusion Injury Influences Hyaluronan Homeostasis in the Rat Brain. Int J Mol Sci. 2025 26
96. Zhou J, Huang WQ, Li C, Wu GY, Li YS, Wen SH. et al. Intestinal ischemia/reperfusion enhances microglial activation and induces cerebral injury and memory dysfunction in rats. Crit Care Med. 2012;40:2438-48
97. Hsieh YH, McCartney K, Moore TA, Thundyil J, Gelderblom M, Manzanero S. et al. Intestinal ischemia-reperfusion injury leads to inflammatory changes in the brain. Shock. 2011;36:424-30
98. Li G, Zhou J, Wei J, Liu B. Dexmedetomidine Ameliorated Cognitive Dysfunction Induced by Intestinal Ischemia Reperfusion in Mice with Possible Relation to the Anti-inflammatory Effect Through the Locus Coeruleus Norepinephrine System. Neurochem Res. 2022;47:3440-53
99. Chen XD, Zhao J, Yang X, Zhou BW, Yan Z, Liu WF. et al. Gut-Derived Exosomes Mediate Memory Impairment After Intestinal Ischemia/Reperfusion via Activating Microglia. Mol Neurobiol. 2021;58:4828-41
100. Gao X, Lin C, Feng Y, You Y, Jin Z, Li M. et al. Akkermansia muciniphila-derived small extracellular vesicles attenuate intestinal ischemia-reperfusion-induced postoperative cognitive dysfunction by suppressing microglia activation via the TLR2/4 signaling. Biochim Biophys Acta Mol Cell Res. 2024;1871:119630
101. Kärkkäinen JM. Acute mesenteric ischemia in elderly patients. Expert Rev Gastroenterol Hepatol. 2016;10:985-8
102. Kassahun WT, Schulz T, Richter O, Hauss J. Unchanged high mortality rates from acute occlusive intestinal ischemia: six year review. Langenbecks Arch Surg. 2008;393:163-71
103. Aschoff AJ, Stuber G, Becker BW, Hoffmann MH, Schmitz BL, Schelzig H. et al. Evaluation of acute mesenteric ischemia: accuracy of biphasic mesenteric multi-detector CT angiography. Abdom Imaging. 2009;34:345-57
104. Collins JD. MR Imaging of the Mesenteric Vasculature. Radiol Clin North Am. 2020;58:797-813
105. Carlos RC, Stanley JC, Stafford-Johnson D, Prince MR. Interobserver variability in the evaluation of chronic mesenteric ischemia with gadolinium-enhanced MR angiography. Acad Radiol. 2001;8:879-87
106. Laissy JP, Trillaud H, Douek P. MR angiography: noninvasive vascular imaging of the abdomen. Abdom Imaging. 2002;27:488-506
107. Wasser MN, Geelkerken RH, Kouwenhoven M, van Bockel JH, Hermans J, Schultze Kool LJ. et al. Systolically gated 3D phase contrast MRA of mesenteric arteries in suspected mesenteric ischemia. J Comput Assist Tomogr. 1996;20:262-8
108. Ginsburg M, Obara P, Lambert DL, Hanley M, Steigner ML, Camacho MA. et al. ACR Appropriateness Criteria(®) Imaging of Mesenteric Ischemia. J Am Coll Radiol. 2018;15:S332-s40
109. Perko MJ. Duplex ultrasound for assessment of superior mesenteric artery blood flow. Eur J Vasc Endovasc Surg. 2001;21:106-17
110. Zwolak RM, Fillinger MF, Walsh DB, LaBombard FE, Musson A, Darling CE. et al. Mesenteric and celiac duplex scanning: a validation study. J Vasc Surg. 1998;27:1078-87 discussion 88
111. Moneta GL, Lee RW, Yeager RA, Taylor LM Jr, Porter JM. Mesenteric duplex scanning: a blinded prospective study. J Vasc Surg. 1993;17:79-84 discussion 5-6
112. Wolf EL, Sprayregen S, Bakal CW. Radiology in intestinal ischemia. Plain film, contrast, and other imaging studies. Surg Clin North Am. 1992;72:107-24
113. Chiu YH, Huang MK, How CK, Hsu TF, Chen JD, Chern CH. et al. D-dimer in patients with suspected acute mesenteric ischemia. Am J Emerg Med. 2009;27:975-9
114. Powell A, Armstrong P. Plasma biomarkers for early diagnosis of acute intestinal ischemia. Semin Vasc Surg. 2014;27:170-5
115. Block T, Nilsson TK, Björck M, Acosta S. Diagnostic accuracy of plasma biomarkers for intestinal ischaemia. Scand J Clin Lab Invest. 2008;68:242-8
116. Gün B, Yolcu S, Değerli V, Elçin G, Tomruk Ö, Erdur B. et al. Multi-detector angio-CT and the use of D-dimer for the diagnosis of acute mesenteric ischemia in geriatric patients. Ulus Travma Acil Cerrahi Derg. 2014;20:376-81
117. Cronk DR, Houseworth TP, Cuadrado DG, Herbert GS, McNutt PM, Azarow KS. Intestinal fatty acid binding protein (I-FABP) for the detection of strangulated mechanical small bowel obstruction. Curr Surg. 2006;63:322-5
118. Thuijls G, van Wijck K, Grootjans J, Derikx JP, van Bijnen AA, Heineman E. et al. Early diagnosis of intestinal ischemia using urinary and plasma fatty acid binding proteins. Ann Surg. 2011;253:303-8
119. Murray MJ, Gonze MD, Nowak LR, Cobb CF. Serum D(-)-lactate levels as an aid to diagnosing acute intestinal ischemia. Am J Surg. 1994;167:575-8
120. Poeze M, Froon AH, Greve JW, Ramsay G. D-lactate as an early marker of intestinal ischaemia after ruptured abdominal aortic aneurysm repair. Br J Surg. 1998;85:1221-4
121. Brillantino A, Iacobellis F, Renzi A, Nasti R, Saldamarco L, Grillo M. et al. Diagnostic value of arterial blood gas lactate concentration in the different forms of mesenteric ischemia. Eur J Trauma Emerg Surg. 2018;44:265-72
122. Kintu-Luwaga R, Galukande M, Owori FN. Serum lactate and phosphate as biomarkers of intestinal ischemia in a Ugandan tertiary hospital: a cross-sectional study. Int J Emerg Med. 2013;6:44
123. Ferrada P, Callcut R, Bauza G, O'Bosky KR, Luo-Owen X, Mansfield NJ. et al. Pneumatosis Intestinalis Predictive Evaluation Study: A multicenter epidemiologic study of the American Association for the Surgery of Trauma. J Trauma Acute Care Surg. 2017;82:451-60
124. Gunduz A, Turedi S, Mentese A, Karahan SC, Hos G, Tatli O. et al. Ischemia-modified albumin in the diagnosis of acute mesenteric ischemia: a preliminary study. Am J Emerg Med. 2008;26:202-5
125. Kulu R, Akyildiz H, Akcan A, Oztürk A, Sozuer E. Plasma citrulline measurement in the diagnosis of acute mesenteric ischaemia. ANZ J Surg. 2017;87:E57-e60
126. Cossé C, Sabbagh C, Fumery M, Zogheib E, Mauvais F, Browet F. et al. Serum procalcitonin correlates with colonoscopy findings and can guide therapeutic decisions in postoperative ischemic colitis. Dig Liver Dis. 2017;49:286-90
127. Markogiannakis H, Memos N, Messaris E, Dardamanis D, Larentzakis A, Papanikolaou D. et al. Predictive value of procalcitonin for bowel ischemia and necrosis in bowel obstruction. Surgery. 2011;149:394-403
128. Nagata J, Kobayashi M, Nishikimi N, Komori K. Serum procalcitonin (PCT) as a negative screening test for colonic ischemia after open abdominal aortic surgery. Eur J Vasc Endovasc Surg. 2008;35:694-7
129. Sgourakis G, Papapanagiotou A, Kontovounisios C, Karamouzis MV, Lanitis S, Konstantinou C. et al. The value of plasma neurotensin and cytokine measurement for the detection of bowel ischaemia in clinically doubtful cases: a prospective study. Exp Biol Med (Maywood). 2013;238:874-80
130. Sutherland F, Cunningham H, Pontikes L, Parsons L, Klassen J. Elevated serum interleukin 6 levels in patients with acute intestinal ischemia. Hepatogastroenterology. 2003;50:419-21
131. Yamamoto T, Umegae S, Kitagawa T, Matsumoto K. The value of plasma cytokine measurement for the detection of strangulation in patients with bowel obstruction: a prospective, pilot study. Dis Colon Rectum. 2005;48:1451-9
132. Aktimur R, Cetinkunar S, Yildirim K, Aktimur SH, Ugurlucan M, Ozlem N. Neutrophil-to-lymphocyte ratio as a diagnostic biomarker for the diagnosis of acute mesenteric ischemia. Eur J Trauma Emerg Surg. 2016;42:363-8
133. Woodford EP, Woodford HM, Hort AR, Pang TC, Lam VWT, Nahm CB. Neutrophil-lymphocyte ratio and platelet-lymphocyte ratio use in detecting bowel ischaemia in adhesional small bowel obstruction. ANZ J Surg. 2022;92:2915-20
134. Schellekens D, Reisinger KW, Lenaerts K, Hadfoune M, Olde Damink SW, Buurman WA. et al. SM22 a Plasma Biomarker for Human Transmural Intestinal Ischemia. Ann Surg. 2018;268:120-6
135. Gearhart SL, Delaney CP, Senagore AJ, Banbury MK, Remzi FH, Kiran RP. et al. Prospective assessment of the predictive value of alpha-glutathione S-transferase for intestinal ischemia. Am Surg. 2003;69:324-9 discussion 9
136. Liu M, Wen H, Zuo L, Song X, Geng Z, Ge S. et al. Bryostatin-1 attenuates intestinal ischemia/reperfusion-induced intestinal barrier dysfunction, inflammation, and oxidative stress via activation of Nrf2/HO-1 signaling. Faseb j. 2023;37:e22948
137. Gendy A, Soubh A, Al-Mokaddem A, Kotb El-Sayed M. Dimethyl fumarate protects against intestinal ischemia/reperfusion lesion: Participation of Nrf2/HO-1, GSK-3β and Wnt/β-catenin pathway. Biomed Pharmacother. 2021;134:111130
138. Shen T, Wang X, Zhang J, Lin Y, Cai L, Deng K. et al. FSP1 reduces exogenous coenzyme Q10 and inhibits ferroptosis to alleviate intestinal ischemia-reperfusion injury. J Adv Res. 2025
139. Li XY, Xie JY, Yin YT, Weng XG, Huang ZY, Wang LS. et al. Obacunone inhibits RIPK1/RIPK3/MLKL-mediated necroptosis by suppressing mitochondrial ROS and MIF signaling in intestinal ischemia-reperfusion injury. Int Immunopharmacol. 2026;168:115901
140. Akhigbe RE, Adedamola Aminat BO, Akhigbe TM, Hamed MA. Glutamine Alleviates I/R-Induced Intestinal Injury and Dysmotility Via the Downregulation of Xanthine Oxidase/Uric Acid Signaling and Lactate Generation in Wistar Rats. J Surg Res. 2024;295:431-41
141. Turan I, Ozacmak HS, Ozacmak VH, Barut F, Araslı M. Agmatine attenuates intestinal ischemia and reperfusion injury by reducing oxidative stress and inflammatory reaction in rats. Life Sci. 2017;189:23-8
142. Gu L, Wang F, Wang Y, Sun D, Sun Y, Tian T. et al. Naringin protects against inflammation and apoptosis induced by intestinal ischemia-reperfusion injury through deactivation of cGAS-STING signaling pathway. Phytother Res. 2023;37:3495-507
143. Li Y, Wen S, Yao X, Liu W, Shen J, Deng W. et al. MicroRNA-378 protects against intestinal ischemia/reperfusion injury via a mechanism involving the inhibition of intestinal mucosal cell apoptosis. Cell Death Dis. 2017;8:e3127
144. Feng D, Wang Z, Zhao Y, Li Y, Liu D, Chen Z. et al. circ-PRKCB acts as a ceRNA to regulate p66Shc-mediated oxidative stress in intestinal ischemia/reperfusion. Theranostics. 2020;10:10680-96
145. Chassin C, Hempel C, Stockinger S, Dupont A, Kübler JF, Wedemeyer J. et al. MicroRNA-146a-mediated downregulation of IRAK1 protects mouse and human small intestine against ischemia/reperfusion injury. EMBO Mol Med. 2012;4:1308-19
146. Liu L, Yao J, Li Z, Zu G, Feng D, Li Y. et al. miR-381-3p knockdown improves intestinal epithelial proliferation and barrier function after intestinal ischemia/reperfusion injury by targeting nurr1. Cell Death Dis. 2018;9:411
147. Zhang W, Zhou B, Yang X, Zhao J, Hu J, Ding Y. et al. Exosomal circEZH2_005, an intestinal injury biomarker, alleviates intestinal ischemia/reperfusion injury by mediating Gprc5a signaling. Nat Commun. 2023;14:5437
148. Liu KX, Li YS, Huang WQ, Chen SQ, Wang ZX, Liu JX. et al. Immediate postconditioning during reperfusion attenuates intestinal injury. Intensive Care Med. 2009;35:933-42
149. Wen SH, Li Y, Li C, Xia ZQ, Liu WF, Zhang XY. et al. Ischemic postconditioning during reperfusion attenuates intestinal injury and mucosal cell apoptosis by inhibiting JAK/STAT signaling activation. Shock. 2012;38:411-9
150. Liu KX, Li C, Li YS, Yuan BL, Xu M, Xia Z. et al. Proteomic analysis of intestinal ischemia/reperfusion injury and ischemic preconditioning in rats reveals the protective role of aldose reductase. Proteomics. 2010;10:4463-75
151. Liu SZ, He XM, Zhang X, Zeng FC, Wang F, Zhou XY. Ischemic Preconditioning-Induced SOCS-1 Protects Rat Intestinal Ischemia Reperfusion Injury via Degradation of TRAF6. Dig Dis Sci. 2017;62:105-14
152. De Luca M, Aiuti A, Cossu G, Parmar M, Pellegrini G, Robey PG. Advances in stem cell research and therapeutic development. Nat Cell Biol. 2019;21:801-11
153. Shi Y, Zhang X, Wan Z, Liu X, Chen F, Zhang J. et al. Mesenchymal stem cells against intestinal ischemia-reperfusion injury: a systematic review and meta-analysis of preclinical studies. Stem Cell Res Ther. 2022;13:216
154. Jiang H, Qu L, Li Y, Gu L, Shi Y, Zhang J. et al. Bone marrow mesenchymal stem cells reduce intestinal ischemia/reperfusion injuries in rats. J Surg Res. 2011;168:127-34
155. Zhang G, Wan Z, Liu Z, Liu D, Zhao Z, Leng Y. Exosomes Derived from BMSCs Ameliorate Intestinal Ischemia-Reperfusion Injury by Regulating miR-144-3p-Mediated Oxidative Stress. Dig Dis Sci. 2022;67:5090-106
156. Inan M, Bakar E, Cerkezkayabekir A, Sanal F, Ulucam E, Subaşı C. et al. Mesenchymal stem cells increase antioxidant capacity in intestinal ischemia/reperfusion damage. J Pediatr Surg. 2017;52:1196-206
157. Liu Z, Zhao Z, Xiao Z, Li M, Wang X, Huang Y. et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells regulate SREBF2/HMGB1 axis by transporting miR-378a-3p to inhibit ferroptosis in intestinal ischemia-reperfusion injury. Cell Death Discov. 2025;11:223
158. Liu L, He YR, Liu SJ, Hu L, Liang LC, Liu DL. et al. Enhanced Effect of IL-1β-Activated Adipose-Derived MSCs (ADMSCs) on Repair of Intestinal Ischemia-Reperfusion Injury via COX-2-PGE(2) Signaling. Stem Cells Int. 2020;2020:2803747
159. Ye K, Peng QF, Zhang ZG, Li X, Xue HB, Guo H. et al. Resveratrol primed adipose stem cell-derived exosomes alleviate intestinal ischemia reperfusion injury by regulating macrophage polarization. Int Immunopharmacol. 2025;165:115481
160. Chang CL, Sung PH, Sun CK, Chen CH, Chiang HJ, Huang TH. et al. Protective effect of melatonin-supported adipose-derived mesenchymal stem cells against small bowel ischemia-reperfusion injury in rat. J Pineal Res. 2015;59:206-20
161. Wang S, Yu L, Sun M, Mu S, Wang C, Wang D. et al. The therapeutic potential of umbilical cord mesenchymal stem cells in mice premature ovarian failure. Biomed Res Int. 2013;2013:690491
162. Gao Y, Chen H, Cang X, Chen H, Di Y, Qi J. et al. Transplanted hair follicle mesenchymal stem cells alleviated small intestinal ischemia-reperfusion injury via intrinsic and paracrine mechanisms in a rat model. Front Cell Dev Biol. 2022;10:1016597
163. Duranti S, Vivo V, Zini I, Milani C, Mangifesta M, Anzalone R. et al. Bifidobacterium bifidum PRL2010 alleviates intestinal ischemia/reperfusion injury. PLoS One. 2018;13:e0202670
164. Wang B, Huang Q, Zhang W, Li N, Li J. Lactobacillus plantarum prevents bacterial translocation in rats following ischemia and reperfusion injury. Dig Dis Sci. 2011;56:3187-94
165. Salim SY, Young PY, Lukowski CM, Madsen KL, Sis B, Churchill TA. et al. VSL#3 probiotics provide protection against acute intestinal ischaemia/reperfusion injury. Benef Microbes. 2013;4:357-65
166. Li T, Cai H, Qiu C, Zhang J, Zhou S, Xian W. et al. Lactobacillus amplifies DHAMaR1 conversion to attenuate intestinal ischemia-reperfusion injury via decreasing pyroptosis. Front Immunol. 2025;16:1712761
167. Ascher S, Wilms E, Pontarollo G, Formes H, Bayer F, Müller M. et al. Gut Microbiota Restricts NETosis in Acute Mesenteric Ischemia-Reperfusion Injury. Arterioscler Thromb Vasc Biol. 2020;40:2279-92
168. Aguilar-Nascimento JE, Salomão AB, Nochi RJ Jr, Nascimento M, Neves Jde S. Intraluminal injection of short chain fatty acids diminishes intestinal mucosa injury in experimental ischemia-reperfusion. Acta Cir Bras. 2006;21:21-5
169. Qiao Y, Qian J, Lu Q, Tian Y, Chen Q, Zhang Y. Protective effects of butyrate on intestinal ischemia-reperfusion injury in rats. J Surg Res. 2015;197:324-30
170. Deng F, Hu JJ, Lin ZB, Sun QS, Min Y, Zhao BC. et al. Gut microbe-derived milnacipran enhances tolerance to gut ischemia/reperfusion injury. Cell Rep Med. 2023;4:100979
171. Liang Y, Sheng R, Hu Y, Huang D, Li X, Deng H. et al. Propionate intervenes mast cell function to alleviate intestinal ischemia-reperfusion injury by regulating AhR/Notch1 pathway. Microb Pathog. 2026;210:108204
172. Wang F, Wang X, Wang C, Yan W, Xu J, Song Z. et al. Gut microbiota-derived glutathione from metformin treatment alleviates intestinal ferroptosis induced by ischemia/reperfusion. BMC Med. 2025;23:285
173. Huang W, Yan Y, Wu M, Hu J, Zhao J, Chen X. et al. Preoperative fasting confers protection against intestinal ischaemia/reperfusion injury by modulating gut microbiota and their metabolites in a mouse model. Br J Anaesth. 2022;128:501-12
174. Zhang FL, Chen XW, Wang YF, Hu Z, Zhang WJ, Zhou BW. et al. Microbiota-derived tryptophan metabolites indole-3-lactic acid is associated with intestinal ischemia/reperfusion injury via positive regulation of YAP and Nrf2. J Transl Med. 2023;21:264
175. Jia Y, He M, Wang F, Zhan Y, Deng Q, Shen J. et al. Indole-3-lactic acid protects the gut vascular barrier following intestinal ischemia injury through AhR/Nrf2/STAT3 mediated claudin 2 downregulation. Cell Commun Signal. 2025;23:447
176. Lagoa CE, de Figueiredo LF, Cruz RJ Jr, Silva E, Rocha e Silva M. Effects of volume resuscitation on splanchnic perfusion in canine model of severe sepsis induced by live Escherichia coli infusion. Crit Care. 2004;8:R221-8
177. Corcos O, Nuzzo A. Gastro-intestinal vascular emergencies. Best Pract Res Clin Gastroenterol. 2013;27:709-25
178. Nuzzo A, Maggiori L, Paugam-Burtz C, Cazals-Hatem D, Ronot M, Huguet A. et al. Oral Antibiotics Reduce Intestinal Necrosis in Acute Mesenteric Ischemia: A Prospective Cohort Study. Am J Gastroenterol. 2019;114:348-51
179. Corcos O, Castier Y, Sibert A, Gaujoux S, Ronot M, Joly F. et al. Effects of a multimodal management strategy for acute mesenteric ischemia on survival and intestinal failure. Clin Gastroenterol Hepatol. 2013;11:158-65.e2
180. Pedersoli F, Schönau K, Schulze-Hagen M, Keil S, Isfort P, Gombert A. et al. Endovascular Revascularization with Stent Implantation in Patients with Acute Mesenteric Ischemia due to Acute Arterial Thrombosis: Clinical Outcome and Predictive Factors. Cardiovasc Intervent Radiol. 2021;44:1030-8
181. Heiss P, Loewenhardt B, Manke C, Hellinger A, Dietl KH, Schlitt HJ. et al. Primary percutaneous aspiration and thrombolysis for the treatment of acute embolic superior mesenteric artery occlusion. Eur Radiol. 2010;20:2948-58
182. Yanar H, Taviloglu K, Ertekin C, Ozcinar B, Yanar F, Guloglu R. et al. Planned second-look laparoscopy in the management of acute mesenteric ischemia. World J Gastroenterol. 2007;13:3350-3
183. Zhang J, Duan ZQ, Song QB, Luo YW, Xin SJ, Zhang Q. Acute mesenteric venous thrombosis: a better outcome achieved through improved imaging techniques and a changed policy of clinical management. Eur J Vasc Endovasc Surg. 2004;28:329-34
184. Morasch MD, Ebaugh JL, Chiou AC, Matsumura JS, Pearce WH, Yao JS. Mesenteric venous thrombosis: a changing clinical entity. J Vasc Surg. 2001;34:680-4
185. Calame P, Winiszewski H, Doussot A, Malakhia A, Grillet F, Verdot P. et al. Evaluating the Risk of Irreversible Intestinal Necrosis Among Critically Ill Patients with Nonocclusive Mesenteric Ischemia. Am J Gastroenterol. 2021;116:1506-13
Author contact
Corresponding authors: Jing-Juan Hu and Ke-Xuan Liu, Department of Anesthesiology, Nanfang Hospital, Southern Medical University, 1838 Guangzhou Avenue North, Guangzhou, 510515, China. Electronic address: liukexuan705com; Electronic address: hjjuan0828com.

 Global reach, higher impact
Global reach, higher impact