Impact Factor ISSN: 1449-2288
Int J Biol Sci 2026; 22(8):4116-4135. doi:10.7150/ijbs.131080 This issue Cite
Review
The Role of Exosomes in Offspring Metabolic Programming in Gestational Diabetes: Mechanisms and Potential Applications
1. NHC Key Laboratory of Endocrinology (Peking Union Medical College Hospital), Diabetes Research Center of Chinese Academy of Medical Sciences, Department of Endocrinology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, 100730, China.
2. Department of Endocrinology, Genetics, Metabolism, Beijing Children's Hospital, Capital Medical University, National Center for Children's Health, Beijing, China.
3. Department of Obstetrics and Gynaecology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China.
4. Center for Biomarker Discovery and Validation, National Infrastructures for Translational Medicine (PUMCH), Institute of Clinical Medicine, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China.
# These authors have contributed equally to this work and share first authorship.
Received 2026-1-7; Accepted 2026-3-17; Published 2026-3-30
Abstract
Genetic predisposition and unhealthy lifestyles are well-known contributors to disorders of glucose and lipid metabolism, including type 2 diabetes, obesity, and metabolic dysfunction-associated fatty liver disease. However, these factors alone cannot fully explain the rapidly rising prevalence of these conditions. Emerging evidence highlights the pivotal role of the intrauterine environment in gestational diabetes mellitus (GDM) in shaping epigenetic modifications and metabolic reprogramming, thereby predisposing offspring to long-term metabolic complications. Exosomes have recently been identified as key mediators of maternal-fetal communication. In GDM, both the quantity and cargo (e.g., proteins, miRNAs) of exosomes are altered. These altered exosomes not only contribute to maternal glucose and lipid metabolic abnormalities but also act as a critical vector for transmitting adverse metabolic signals to the offspring. This exosome-mediated communication disrupts placental function and the development of fetal metabolic organs, ultimately programming the offspring for long-term metabolic disorders. In this review, we summarize the characteristic changes of maternal exosomes in GDM and explore the potential mechanism by which exosomes regulate offspring metabolism during maternal-fetal crosstalk. We also propose the possible direction of exosomes in application, providing insights into early-life strategies for the prevention of metabolic diseases.
Keywords: Gestational diabetes mellitus, Exosomes, Maternal-fetal communication, Offspring, Glucose and lipid metabolism
1. Introduction
Gestational diabetes mellitus (GDM) is one of the most common complications in pregnancy [1]. The latest data from the International Diabetes Federation reported that one fetus in six live births is born to GDM mothers [2]. A substantial body of evidence suggests that GDM has the potential to permanently alter metabolic programming and physiology, thus contributing to metabolic disturbances in both mothers and their offspring [3].
The early intrauterine environment can affect the metabolic status in later life, a concept known as the developmental origins of health and disease (DOHaD) theory [4]. Since 1986, numerous studies have underscored the pivotal role of the intrauterine period in cellular growth, differentiation, and organogenesis, showing that maternal and early-life exposures are essential for normal fetal growth and the long-term development of offspring [5-8]. The potential mechanisms underlying the effects of adverse intrauterine environment, such as hyperglycemia in GDM, on metabolism during pregnancy and offspring metabolism include abnormal placental function [9], epigenetic and metabolic reprogramming [10]. However, the relevant mechanisms are not completely clear, and early intervention measures still need further research. In this context, increasing attention has been directed toward soluble and vesicle-mediated signaling pathways that can cross the placental barrier and deliver regulatory molecules from the mother to the fetus, among which exosomes have emerged as particularly promising candidates.
As a class of extracellular vesicles (EVs), exosomes can stably carry miRNAs, mRNAs, proteins, and lipids in biofluids [11]. These cargo-enriched exosomes are now recognized as critical mediators of intercellular communication, thereby directly modulating gene expression and signaling in recipient cells [12]. In vivo and in vitro studies indicate that blocking exosome biogenesis or release can attenuate inter-tissue signal transmission (e.g., adipose-derived circulating miRNAs that regulate the liver and muscle), suggesting that exosomes may play causal or amplifying roles in metabolic programming [13-15].
Pregnancy itself is a state of dynamic intercellular communication, during which both the concentration and molecular profile of circulating exosomes are remodeled [16]. Moreover, the characteristic pathological milieu of GDM can markedly alter the quantity, cargo composition, and biological function of exosomes derived from maternal tissues [17-19]. These GDM-associated changes may represent a key pathway through which adverse metabolic effects are transmitted to the offspring. Indeed, studies in mouse models have demonstrated that maternal exosomes can cross the placenta and enter the fetal circulation [20]. Consequently, exosomes derived from GDM mothers can modulate placental function and key aspects of fetal metabolic programming, such as metabolic organ maturation, fetal growth, and body composition [21-24]. Furthermore, exosomes are easily detectable in blood and can serve as potential targets for therapeutic intervention. Therefore, considering exosomes as a measurable and potentially modifiable mode of interorgan communication can complement and deepen our understanding of GDM-maternal-fetal interactions and guide potential early diagnostic and interventional strategies.
In this review, we summarize the characteristic changes of maternal exosomes under GDM, explore the transgenerational role of exosomes in offspring metabolism during maternal-fetal crosstalk, and highlight their potential therapeutic applications. These insights may advance our understanding of the pathogenesis and early intervention during the critical therapeutic window for metabolic disorders.
2. Overview of Exosomes in Pregnancy
EVs are lipid bilayer-enclosed particles actively secreted by cells, originating from various compartments, including endosomes and the plasma membrane. They are widely distributed in bodily fluids such as blood, saliva, and urine, as well as in cell culture supernatants [11]. According to the biological pathway, size and release mechanism, EVs can be divided into three main types: exosomes (30-150 nm, classified as small EVs (sEVs), derived from the fusion of multivesicular bodies (MVBs) and plasma membrane), microvesicles (100-1000 nm, produced by outward budding of plasma membrane) and apoptotic bodies (1000-5000 nm, formed in the process of programmed cell death) [25-27]. EVs transport diverse functional molecules, including proteins, lipids, RNA/DNA species, and bioactive enzymes [28], with release occurring across nearly all cell types [29]. This ubiquitous distribution underscores their crucial role in cellular communication mechanisms.
2.1 Exosome biogenesis, uptake, and characterization
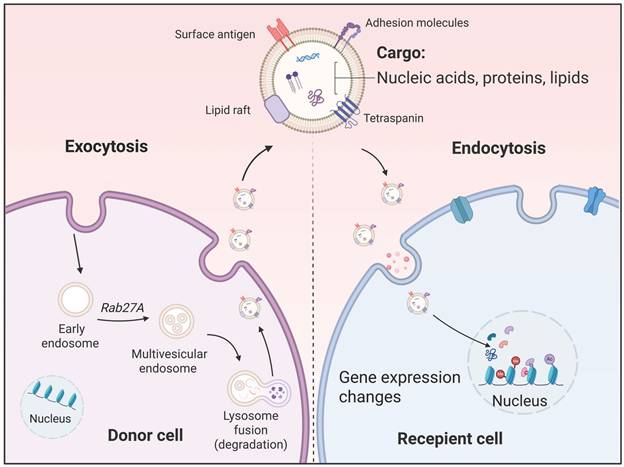
As a crucial subcategory of EVs, exosomes serve as vital messengers in both physiological and pathological processes. The biological generation of exosomes begins with the invagination of the cell membrane, forming an early endosome. Upon maturation into a late endosome, the membrane undergoes internal budding to encapsulate cytoplasmic contents, creating an MVB packed with numerous intracellular vesicles [30]. Ultimately, this MVB fuses with the cell membrane, releasing its vesicles into the extracellular environment, thus becoming exosomes [31]. The lipid bilayer of exosomes effectively shields and protects their cargo from degradation. Through bodily fluid circulation, exosomes are transported to local or distant target cells, where they deliver their cargo via membrane fusion, endocytosis, or ligand-receptor interactions [27]. These molecular messages subsequently modulate the function of recipient cells, playing critical roles in physiological processes and pathological conditions [32, 33] (Figure 1).
The biological generation and uptake of exosomes. The biological generation of exosomes begins with the invagination of the cell membrane forming an early endosome. Upon maturation into a late endosome, the membrane undergoes internal budding to encapsulate cytoplasmic contents, creating a polycentric vesicle body packed with numerous intracellular vesicles. Ultimately, this polycentric vesicle body fuses with the cell membrane, releasing its vesicles into the extracellular environment, thus becoming exosomes. Through fluid circulation, exosomes deliver their cargo to local or distant target cells via membrane fusion, endocytosis, or ligand-receptor interactions.
Currently, exosomes are commonly isolated from blood (including plasma and serum), urine, and cell culture conditioned medium [18, 34]. Standard isolation techniques include differential (ultra)centrifugation, density gradient centrifugation, size-exclusion chromatography, and others [35]. To enhance the yield, these methods are often combined with ultrafiltration for concentration. The purified exosomes are then characterized using techniques like electron microscopy and nanoparticle tracking analysis (NTA) to confirm their size and morphology. Furthermore, the presence of characteristic exosomal surface markers (e.g., CD63, CD81, CD9, TSG101) is typically verified by western blot or flow cytometry [36]. Due to their critical role in pathogenesis, along with their high abundance and detectable composition, exosomes and their cargo have emerged as a highly promising source of biomarkers for disease diagnosis and prevention [37].
2.2 Exosome transport in pregnancy
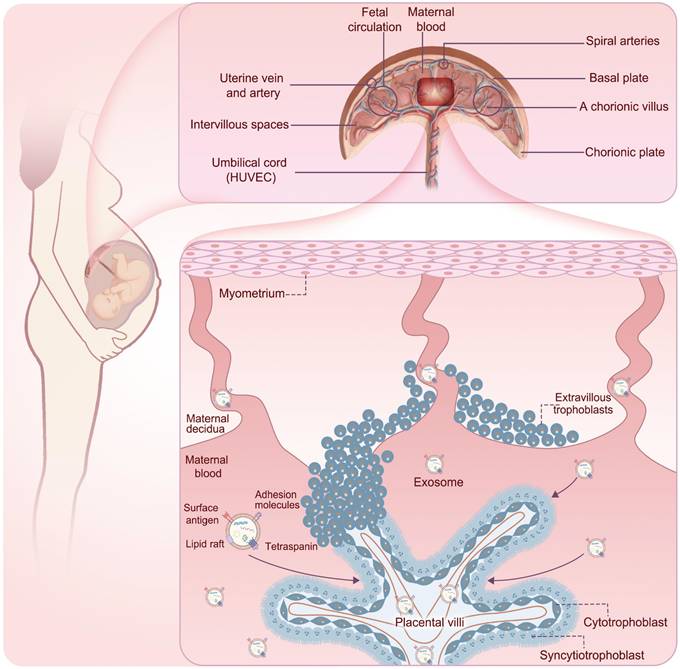
During pregnancy, circulating exosome concentrations in healthy pregnant women increase approximately 20- to 50-fold compared to those in non-pregnant women and continue to increase throughout gestation [16, 38]. This range reflects differences in isolation methods, quantification techniques, and gestational timing across studies. These vesicles contribute to processes such as embryo implantation, placental function, and modulation of the maternal immune system [39], demonstrating their regulatory role in cell-cell communication. Furthermore, the placental barrier is not a static wall but a dynamic interface where maternal exosomes can be selectively transferred to the fetal side [40-42]. Circulating maternal exosomes first encounter the apical surface of the syncytiotrophoblast, the outermost cell layer of the chorionic villi. At this interface, exosomes bind to trophoblast membranes through adhesion molecules and receptor-ligand interactions involving, for example, tetraspanins, integrins, and heparan sulfate proteoglycans, and are internalized predominantly via active endocytic pathways, including clathrin- and caveolin-dependent endocytosis, macropinocytosis, and phagocytosis [41, 43]. Once inside trophoblast cells, exosomes enter the endosomal system, where their cargos may be integrated into placental signaling networks or sorted into vesicles destined for basolateral exocytosis. Through this transcytotic route, intact exosomes and their released cargos can be released into the villous stroma and fetal capillary compartment, thereby reaching the fetal circulation [41, 43]. Experimental mouse models using fluorescently labeled or genetically engineered sEVs have provided direct in vivo evidence that vesicles administered on the maternal side can cross the placenta, be detected in fetal tissues, and induce functional changes, demonstrating maternal-fetal transfer of exosomal cargo across the placental barrier [20, 44]. Fetal endothelial and parenchymal cells, such as hepatocytes [45], pancreatic β cells [46], and adipocytes [47], can further take up these vesicles via similar receptor-mediated and endocytic mechanisms, allowing maternal exosomes to deliver regulatory nucleic acids and proteins to fetal target tissues and potentially modify gene expression, epigenetic marks, and metabolic pathways that contribute to fetal metabolic programming [44] (Figure 2).
The process of exosomes crossing the placenta into the fetus. Circulating maternal exosomes first encounter the apical surface of the syncytiotrophoblast, the outermost cell layer of the chorionic villi. At this interface, exosomes are internalized predominantly via active endocytic pathways. Once inside trophoblast cells, exosomes enter the endosomal system, where their cargos may be integrated into placental signaling networks or sorted into vesicles destined for basolateral exocytosis. Through this transcytotic route, intact exosomes and their released cargos can be released into the villous stroma and fetal capillary compartment, thereby reaching the fetal circulation.
3. Exosomes in GDM
In GDM, the characteristic pathological milieu—hyperglycemia, insulin resistance, oxidative stress, and low-grade inflammation—profoundly alters exosome secretion by the placenta, adipose tissue, endothelial cells, and other maternal tissues [17-19]. Specifically, hyperglycemia and insulin resistance, as key pathological stimuli, activate signaling pathways such as PKC/NF-κB and HIF-1α in placental trophoblast cells, upregulating the expression of exosome biogenesis-related proteins (e.g., Rab27a), thereby significantly increasing the release of placental-derived exosomes [48, 49]. Meanwhile, in states of insulin resistance and lipotoxicity, endoplasmic reticulum stress and local inflammatory responses are also activated in adipose tissue, promoting the secretion of exosomes carrying specific miRNAs and adipokines by adipocytes and macrophages [50, 51]. Endothelial EVs show reduced vasoprotective miRNAs and increased procoagulant cargos, thereby leading to vascular dysfunction [52, 53]. As a result, exosomes display characteristic changes in abundance, cargo composition, and bioactivity, which are thought to represent a key route through which adverse metabolic signals are transmitted from mother to offspring [17-19]. Circulating exosomes in women with GDM show higher concentrations and larger mean diameters than those in normoglycemic pregnant women, and their levels correlate positively with maternal glycemia [17]. Functionally, injections of exosomes isolated from the plasma of GDM mothers into mice reduce basal insulin signaling and insulin responsiveness in skeletal muscle [54, 55].
To further elucidate the roles of these vesicles, miRNA sequencing and proteomic analyses have been widely used to identify differentially expressed cargos in GDM-associated exosomes [56, 57], many of which are closely linked to pathways involved in glucose and lipid metabolism and metabolic inflammation. In the following section, we discuss the exosomes in GDM according to their major molecular cargos.
3.1 miRNAs in exosomes from GDM
Among the compositional changes in exosomes, altered miRNA profiles are the most striking. These miRNAs, as key post-transcriptional regulatory factors, are considered major mediators of the metabolic disturbances conveyed by GDM-derived exosomes [58, 59]. Several studies have reported that GDM induces significant changes in exosomal miRNA levels [58-60]. For example, compared with those in the normal glucose tolerance (NGT) group, 9 miRNAs were significantly upregulated and 14 downregulated in exosomes isolated from the GDM group, and the levels of these miRNAs were consistent in the placenta, circulating exosomes, and skeletal muscle [58]. These significantly altered exosomal miRNAs are positively correlated with fasting plasma glucose (FPG) and OGTT levels in GDM mothers [61, 62]. Other studies have reported that significantly altered miRNAs in exosomes from GDM mothers include miR-122-5p [60], miR-92a-3p [63], miR-320b [64], miR-99-5p [65], etc. These miRNAs are transported to target cells and regulate insulin signaling, adipogenesis, placental function, angiogenesis, and other pathways (Table 1).
miRNAs in exosomes from GDM.
| Contents in GDM exosomes | Source | Sample/Model | Trimester of pregnancy | Isolation of exosomes | Characterization of exosomes | Differential expression in GDM | Target/effect | Reference |
|---|---|---|---|---|---|---|---|---|
| miR-122-5p | Serum | Human (Caucasian/White) | 6-15GW | exoRNeasy Serum/Plasma Maxi kit | TEM | ↑ | PPARα↓, Sirt1↓ in the liver (when miR-122-5p is upregulated) | [59] |
| Plasma | Human (Asian) | 10-16GW | Differential ultracentrifugation | TEM; NTA; WB | ↓ | [59, 65] | ||
| Plasma | Human (Asian) | 8-12GW | Ultracentrifugation | TEM; NTA; WB | ↓ | [59, 60] | ||
| miR-132-3p | Serum | Human (Caucasian/White) | 6-15GW | exoRNeasy Serum/Plasma Maxi kit | TEM | ↑ | KLF7↓ in endothelial cells | [59] |
| miR-29a-3p | Serum | Human (Caucasian/White) | 6-15GW | exoRNeasy Serum/Plasma Maxi kit | TEM | ↑ | Sirt1↓, Foxo3↓ in the liver | [59, 75] |
| miR-29b-3p | Serum | Human (Caucasian/White) | 6-15GW | exoRNeasy Serum/Plasma Maxi kit | TEM | ↑ | [59, 75] | |
| miR-182-3p | Serum | Human (Caucasian/White) | 6-15GW | exoRNeasy Serum/Plasma Maxi kit | TEM | ↑ | INSR1↓ in the skeletal muscle | [59, 157] |
| miR-92a-3p | Plasma | Human (Caucasian/White) | <18GW; 22-28GW; 37-40GW | Ultracentrifugation | TEM; NTA; WB | <18GW ↑; 22-28GW↑; 37-40GW↓ | SOCS2↑, NOS2↓ in skeletal muscle cells (when miR-92a-3p is upregulated) | [63] |
| miR-16-2-3p | Plasma | Human (Caucasian/White) | <18GW; 22-28GW; 37-40GW | Ultracentrifugation | TEM; NTA; WB | <18GW ↑; 22-28GW↑; 37-40GW↓ | ACADM↓ in vascular cells (when miR-16-2-3p is upregulated) | [63, 158] |
| miR-16-5p | Urine (PLAP+) | Human (Caucasian/White) | 8-20GW; 24-28GW; 32-39GW | Urine Exosome Purification kit | TEM; WB | 8-20GW (-); 24-28GW↑; 32-39GW (-) | IRS1↓, IRS2↓ in the liver | [78] |
| miR-1299 | Plasma | Human (Asian) | 24-28GW | Differential ultracentrifugation | TEM; NTA; WB | ↑ | STAT3/FAM3A↓ in hepatocytes | [159] |
| miR-423-5p | Plasma | Human (Asian) | 10-16GW | Differential ultracentrifugation | TEM; NTA; WB | ↑ | IGF1R↓, GYS1↓ in HepG2 cells | [65] |
| miR-148a-3p | Plasma | Human (Asian) | 10-16GW | Differential ultracentrifugation | TEM; NTA; WB | ↓ | TGFB2↑, FGF2↑ in retinal microvascular endothelial cells | [65, 160] |
| miR-192-5p | Plasma | Human (Asian) | 10-16GW | Differential ultracentrifugation | TEM; NTA; WB | ↓ | SCD-1↑ in the liver | [65, 161] |
| miR-99a-5p | Plasma | Human (Asian) | 10-16GW | Differential ultracentrifugation | TEM; NTA; WB | ↓ | mTOR↑, NLRP3↑ in macrophages | [65, 162] |
| miR-99b-5p | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↑ | mTOR↓ in the kidney | [58, 163] |
| miR-125a-3p | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↑ | Adipogenesis↑ | [58, 164] |
| miR-197-3p | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↑ | ROBO1↓ in the UVECs | [58, 165] |
| miR-22-3p | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↑ | GLUT4↑ in the placenta | [58, 166] |
| miR-200-3p | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↑ | TGF-β2/Smad↓ in retina tissue | [58, 167] |
| miR-101-3p | Breast milk | Human (Asian) | Lactation | Ultracentrifugation | TEM; NTA; WB | ↑ | mTOR↓ in hepatocytes | [168] |
| miR-222-3p | Plasma | Human (Caucasian/White) | Late gestation | ExoQuick PlExoQuick asma prep and Exosome precipitation kit | WB | ↑ | ER-α↓, GLUT4↓ in the adipose tissue (when miR-222-3p is upregulated) | [62, 78] |
| Urine (PLAP+) | Human (Caucasian/White) | 8-20GW; 24-28GW; 32-39GW | Urine Exosome Purification kit | TEM; WB | 8-20GW (-); 24-28GW↑; 32-39GW↓ | [78] | ||
| miR-409-3p | Plasma | Human (Caucasian/White) | Late gestation | Plasma prep and Exosome precipitation kit | WB | ↑ | Positively correlated with FPG and HbA1C | [62] |
| has-miR-451a | Placenta | Human (Caucasian/White) | At term | Differential ultracentrifugation+density gradient centrifugation | TEM; NTA; WB | ↓ | Glycerol kinase↑ in the liver | [58, 169] |
| Let-7c-5p | Plasma | Human (Asian) | 8-12GW | Ultracentrifugation | TEM; NTA; WB | ↓ | Interacts with NRG4-ERBB4 in the liver | [60] |
| miR-27a-5p | Plasma | Human (Asian) | 8-12GW | Ultracentrifugation | TEM; NTA; WB | ↓ | PPARγ↑, PI3K/AKT-GLUT4↑ in adipocytes | [60] |
| miR-320b | Placenta | Human (Asian) | At term | Differential centrifugation | TEM; NTA; WB | ↑ | β cell apoptosis↑ | [64] |
| miR-135a-5p | Plasma (PLAP+) | Human (Asian) | 24-28GW | exoEasy Maxi kit | TEM; NTA; WB | ↑ | Sirtuin 1↑, PI3K/AKT↑ in the trophoblast cells | [170] |
| miR-516b-5p | Urine (PLAP+) | Human (Caucasian/White) | 8-20GW; 24-28GW; 32-39GW | Urine Exosome Purification kit | TEM; WB | 8-20GW (-); 24-28GW↑; 32-39GW↓ | Regulates gene expression in the placenta and adipose tissue | [78] |
| miR-517-5p | Urine (PLAP+) | Human (Caucasian/White) | 8-20GW; 24-28GW; 32-39GW | Urine Exosome Purification kit | TEM; WB | 8-20GW (-); 24-28GW↑; 32-39GW↓ | Regulates gene expression in the placenta and adipose tissue | [78] |
| miR-518a-3p | Urine (PLAP+) | Human (Caucasian/White) | 8-20GW; 24-28GW; 32-39GW | Urine Exosome Purification kit | TEM; WB | 8-20GW (-); 24-28GW↑; 32-39GW↓ | Regulates gene expression in the placenta and adipose tissue | [78] |
ACADM: Acyl-CoA Dehydrogenase Medium Chain; ER-α: Estrogen Receptor-alpha; FGF2: Fibroblast Growth Factor 2; Foxo3: Forkhead box O3; FPG: Fasting Plasma Glucose; GDM: Gestational Diabetes Mellitus; GLUT4: Glucose Transporter Type 4; GW: Gestational Weeks; GYS1: Glycogen Synthase 1; HbA1C: Hemoglobin A1C; IGF1R: Insulin-like Growth Factor 1 Receptor; INSR1: Insulin Receptor Substrate 1; IRS1: Insulin Receptor Substrate 1; IRS2: Insulin Receptor Substrate 2; KLF7: Kruppel-like factor 7; mTOR: Mechanistic Target of Rapamycin; NLRP3: NLR Family Pyrin Domain Containing 3; NOS2: Nitric Oxide Synthase 2; NRG4: Neuregulin 4; NTA: Nanoparticle Tracking Analysis; PLAP: Placental Alkaline Phosphatase; PPARα: Peroxisome Proliferator-Activated Receptor Alpha; PPARγ: Peroxisome Proliferator-Activated Receptor Gamma; PI3K/AKT: Phosphoinositide 3-kinase/Protein Kinase B; ROBO1: Roundabout Guidance Receptor 1; SCD-1: Stearoyl-CoA Desaturase-1; Sirt1: Sirtuin 1; SOCS2: Suppressor of Cytokine Signaling 2; STAT3: Signal Transducer and Activator of Transcription 3; TEM: Transmission Electron Microscopy; TGFB2: Transforming Growth Factor Beta 2; WB: Western Blotting.
3.1.1 Blood-derived exosomal miRNAs in GDM
Given their accessibility and dynamic reflection of systemic metabolic changes, blood-derived exosomes have been extensively characterized in the context of GDM. As a highly abundant liver-associated miRNA, miR-122-5p is central in regulating lipid and glucose metabolism [66]. In metabolic disorders, miR-122-5p is frequently observed at abnormally elevated levels [66, 67]. miR-122-5p may affect gluconeogenesis by targeting Glucose 6 phosphatase [68], and also promote hepatic lipid synthesis through regulating the genes including Acly, Mttp, and Srebp-1 [69]. Consistent with its metabolic functions, multiple studies have reported significant alterations of miR-122-5p in GDM exosomes [59, 60, 65]. However, the reported directions of change are inconsistent. For instance, one study reported upregulation of miR-122-5p in GDM serum exosomes during 6-15 gestational weeks (GW) [59], whereas Shen Y et al. found that the expression of miR-122-5p was decreased in plasma exosomes from GDM mothers at 8-12 GW, followed by an increase in late gestation [60]. Another study found that the expression of miR-122-5p in plasma exosomes was downregulated in GDM mothers during 10-16 GW, suggesting a link to subsequent energy metabolism disorders and insulin resistance [65]. These discrepancies may reflect differences in gestational age at sampling, biofluid type (serum vs. plasma), exosome isolation protocols, and detection methods. Future work is needed to reconcile these inconsistencies and clarify the precise role of exosomal miR-122-5p in GDM.
miR-92a-3p has been confirmed to play a crucial role in GDM. Compared with healthy pregnant women, women with GDM exhibit significantly elevated levels of miR-92a expression in peripheral blood [70]. In plasma exosomes from GDM mothers, miR-92a-3p expression was also significantly increased in mid-pregnancy but markedly decreased in late pregnancy [63, 68], a trend may be influenced by gestational age. In function, miR-92a can target KLF2 to increase insulin secretion in β cells [71], and induces suppressor of cytokine signaling 2 (SOCS2) and inhibits the expression of NOS2 in skeletal muscle cells to prevent hyperglycemia by increasing glucose uptake [58, 63]. In addition, as an angiogenesis-related miRNA, the upregulation of miR-92a-3p in GDM may affect placental vascular remodeling and maternal-fetal interface function [72], which is associated with the risk of complications such as placental hemodynamic disorders and macrosomia [73].
The miR-29 family serves as a dual regulator of insulin secretion and signaling. Exosomes secreted by pancreatic β-cells are rich in miR-29, which targets the regulatory subunit p85α of PI3K, leading to insulin resistance [74]. In the liver, miR-29 promotes lipid synthesis by directly targeting Sirt1 and Foxo3 [75]. Placenta-derived exosomes extracted from the plasma of GDM mothers showed significantly elevated levels of both miR-29a-3p and miR-29b-3p, suggesting that the placenta may regulate maternal metabolism through exosomal secretion [59]. In addition, miR-29 can target DNA methyltransferase (DNMT) and participate in epigenetic regulation [59], which may be related to the long-term effects of metabolic programming.
3.1.2 Urine-derived exosomal miRNAs in GDM
Current research on GDM-derived exosomes has predominantly focused on those isolated from blood and placental tissues, with comparatively less attention given to urinary exosomes. This discrepancy is likely attributable to technical challenges in purification and the fact that urine composition is more susceptible to confounding factors such as diet and exercise [76, 77]. Despite these challenges, Herrera-Van Oostdam AS et al. isolated placenta-derived exosomes (PLAP+) from the urine of GDM mothers, revealing that miR-516-5p, miR-517-3p, and miR-518-5p levels were significantly elevated from the second trimester through the third trimester compared to healthy controls [78]. Notably, their shared target genes are predominantly expressed in placental and adipose tissues, suggesting these exosomes may reach these tissues, and placental regulation constitutes an integral component of maternal-fetal communication [78].
In summary, most differentially expressed exosomal miRNAs in GDM are involved in glucose and lipid metabolic pathways, and many show promise as early biomarkers that could improve the prediction and diagnosis of GDM [60, 65]. However, changes in miRNA expression levels may be inconsistent across studies, likely due to small sample sizes, different gestational weeks, lack of pre-pregnancy or early-pregnancy glycemic data, and heterogeneity in clinical diagnostic criteria for GDM. In addition, most studies focused on the effects of exosomes on the GDM mothers. In fact, these exosomes may cross the placental barrier and influence fetal development and metabolism, a possibility that warrants more in-depth investigation in future studies. Furthermore, research on the differential effects of exosomes derived from various subtypes or severities of GDM remains limited, highlighting another critical avenue for future investigation.
3.2 Proteins in exosomes from GDM
Proteomic approaches have been widely applied to characterize the cargo of GDM-derived exosomes, revealing a large number of differentially expressed proteins. These proteins are associated with inflammatory responses [57], cellular stress [79], maternal metabolism, and changes in placental function [80] (Table 2).
Proteins in exosomes from GDM.
| Contents in GDM exosomes | Source | Sample/Model | Trimester of pregnancy | Isolation of exosomes | Characterization of exosomes | Differential expression in GDM | Target/effect | Reference |
|---|---|---|---|---|---|---|---|---|
| Complement C3, C5, C4B, C4BPB and C4BPA | Serum | Human (Caucasian/White) | Late gestation | Differential centrifugation | Dynamic light scattering (DLS); Zeta potential; WB | ↓ | The classical complement/antigen-binding pathway↓ | [79] |
| C7, C9, F12 | Serum | Human (Caucasian/White) | Late gestation | Differential centrifugation | DLS; Zeta potential; WB | ↑ | The complement terminal pathway and associated inflammatory and coagulation processes↑ | [79] |
| BANF1 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↑ | cGAS-STING in the placenta↓ | [83] |
| CARHSP1 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↓ | Gluconeogenesis in the liver↑ | [83] |
| CRYZL1 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↑ | Affect cell apoptosis and oxidative stress | [83] |
| PSG3 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↑ | Regulate insulin secretion | [83] |
| ACPP | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↑ | Regulate immunity | [83] |
| GBP1 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↓ | Associated with endothelial dysfunction | [83] |
| OTUB1 | Placenta (PLAP+) | Human (Caucasian/White) | At term | Ultracentrifugation | NTA; WB | ↓ | Affect cell signal transduction | [83] |
| Thbs1 | ADSCs | Mice (C57BL/6 J) | 12 days of gestation | Differential centrifugation | TEM; NTA; WB | ↑ | Tgfβ/Smad2 in hepatocytes↑ | [18] |
| GDF3 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↑ | The adipocytes enlarge | [85] |
| PROM1 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↑ | Associated with GDM-related macrosomia | [85] |
ACPP: Acid Phosphatase, Prostate; ADSCs: Adipose-derived stem cells; BANF1: Barrier To Autointegration Factor 1; CARHSP1: Calcium Regulated Heat Stable Protein 1; C4BPA: Complement Component 4 Binding Protein Alpha; C4BPB: Complement Component 4 Binding Protein Beta; CRYZL1: Crystallin Zeta Like 1; F12: Coagulation Factor XII; GDF3: Growth Differentiation Factor 3; GBP1: Guanylate Binding Protein 1; GW: Gestational Weeks; ITIH2: Inter-Alpha-Trypsin Inhibitor Heavy Chain 2; NTA: Nanoparticle Tracking Analysis; PLAP: Placental Alkaline Phosphatase; PROM1: Prominin 1; PSG3: Pregnancy Specific Beta-1-Glycoprotein 3; PTGES3: Prostaglandin E Synthase 3; RANBP1: RAN Binding Protein 1; Thbs1: Thrombospondin 1; TREML2: Triggering Receptor Expressed On Myeloid Cells Like 2; TEM: Transmission Electron Microscopy; Tgfβ: Transforming Growth Factor Beta; WB: Western Blotting.
Exosome proteomics revealed the potential role of the complement system and coagulation factors in the pathogenesis of GDM [81]. In serum exosomes from GDM patients, reduced expression of C3, C5, C4B, C4BPB, and C4BPA suggests suppression of the classical complement/antigen-binding pathway, which may be related to insulin resistance [79]. The upregulation of C7, C9 (key components of the membrane attack complex), and F12 (part of the contact system/endogenous coagulation pathway) in GDM exosomes suggests that the complement terminal pathway and associated inflammatory and coagulation processes are abnormally activated [79]. Such an imbalance may promote inflammatory damage in the placental microenvironment and endothelial dysfunction, potentially contributing to preeclampsia and macrosomia [82].
Differentially expressed proteins in the exosomes reflect maternal metabolism and placental functional status. Thrombospondin 1 (Thbs1) protein was highly expressed in adipose-derived exosomes of GDM mice [18]. It interacts with CD36 and Tgfβ receptors, activates Tgfβ/Smad2 signaling pathways, ultimately inducing insulin resistance [18]. The expression of calcium-regulated heat-stable protein 1 (CARHSP1) was downregulated in the placental exosomes of GDM mothers, which may increase gluconeogenesis and affect inflammatory signal transduction, leading to a hyperglycemic state [83, 84]. Other differentially expressed proteins, such as ACPP (which is related to prostaglandin metabolism), CRYZL1, and ANO10, have also been identified, although their precise roles in GDM remain to be elucidated [83].
Together, these data suggest that exosomal proteins in GDM not only reflect maternal metabolic stress and placental dysfunction but may also participate in maternal-fetal communication by carrying protein signals across or modulating the placental barrier, thereby contributing to metabolic programming in the offspring. Moreover, the distinct proteomic signatures of GDM-associated exosomes highlight their potential as minimally invasive biomarkers and as targets for interventions aimed at improving the intrauterine environment.
3.3 Other non-classical contents in the exosomes from GDM
In addition to miRNAs, exosomes derived from GDM pregnancies also carry other classes of non-coding RNAs, including circular RNAs (circRNAs) and long non-coding RNAs (lncRNAs), which have emerged as potential contributors to disease pathology and biomarkers of GDM (Table 3). The expression of hsa_circRNA_0039480 in plasma exosomes of GDM mothers was higher than that in the NGT group at different stages, and was positively correlated with OGTT in the mid-trimester [61]. The lncRNA AC006064.4, which is highly expressed in GDM exosomes, can be used as a predictor of GDM-related macrosomia [85]. Both circRNAs and lncRNAs may function as "molecular sponges" for miRNAs, thereby indirectly regulating the activity of miRNA, so as to participate in regulating metabolic pathways [86]. Nevertheless, research on the underlying mechanisms of these lncRNAs and circRNAs is poorly understood, highlighting the need for more in-depth studies.
Other non-classical contents in exosomes from GDM.
| Contents in GDM exosomes | Source | Sample/Model | Trimester of pregnancy | Isolation of exosomes | Characterization of exosomes | Differential expression in GDM | Target/effect | Reference |
|---|---|---|---|---|---|---|---|---|
| hsa_circRNA_0039480 | Plasma | Human (Asian) | 11-13GW; 24-26GW; 36-41GW | Polymer precipitation | TEM; NTA; WB | ↑ | Positively correlated with OGTT | [61] |
| AC006064.4 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↑ | Associated with GDM-related macrosomia | [85] |
| lnc-HPS6-1:1 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↑ | Associated with GDM-related macrosomia | [85] |
| circ_0014635 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↑ | Associated with GDM-related macrosomia | [85] |
| lnc-ZFHX3-7: 1 | Umbilical cord blood | Human (Asian) | 24-28GW | Ultracentrifugation | NTA | ↓ | Associated with GDM-related macrosomia | [85] |
| Firmicutes | Serum | Human (Asian) | 24-27GW | Ultracentrifugation | TEM; nFCM | ↑ | Inflammation↑ | [89] |
| Bacteroidetes | Serum | Human (Asian) | 24-27GW | Ultracentrifugation | TEM; nFCM | ↓ | Butyrate↓ | [89] |
GW: Gestational Weeks; nFCM: nano-Flow Cytometry; NTA: Nanoparticle Tracking Analysis; OGTT: Oral Glucose Tolerance Test; TEM: Transmission Electron Microscopy; WB: Western Blotting.
Furthermore, changes in the maternal microbiome may be responsible for metabolic diseases [87, 88]. Microbial components in exosomes can regulate the progression of GDM. Through microbiome analysis, Chang CJ et al. found that the ratio of Firmicutes/Bacteroidetes in serum sEVs of GDM mothers increased, which is associated with insulin resistance [89]. The relative abundance of Bacteroidetes decreased, which resulted in reduced butyrate production, reduced insulin sensitivity, and increased inflammation [89, 90]. The enrichment of Gram-negative pathogens in GDM exosomes may lead to metabolic endotoxemia and activation of the inflammatory response to aggravate the inflammatory response, damage the insulin signaling mechanism, and eventually lead to hyperglycemia [89].
In summary, exosomes originating from GDM carry diverse classes of bioactive molecules, including miRNAs, proteins, other non-coding RNAs, and microbiome-related components. These molecules not only mirror maternal metabolic stress, placental dysfunction, and inflammatory status, but also may act as active mediators of maternal-fetal communication by modulating insulin signaling, lipid metabolism, vascular remodeling, and epigenetic programming in target tissues. The multifaceted exosomal signatures observed in GDM therefore provide a mechanistic link between the adverse maternal environment and offspring metabolic risk, while at the same time offering a rich source of minimally invasive biomarkers and potential targets for early intervention.
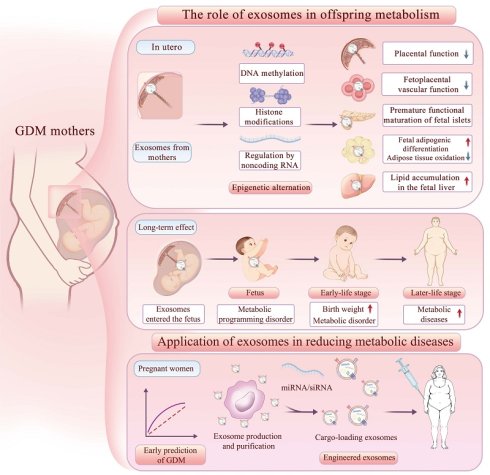
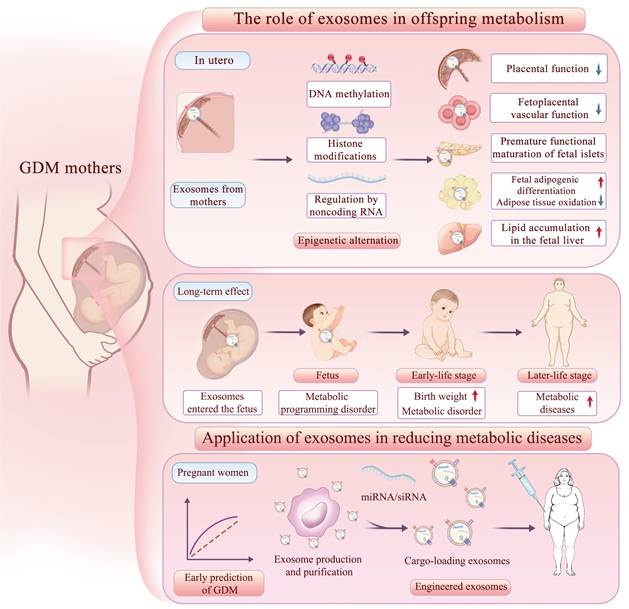
4. The Role of Exosomes in Offspring Metabolism
The DOHaD hypothesis proposes that an adverse intrauterine environment can shape long-term metabolic health in the offspring, largely through epigenetic modifications. Multiple clinical studies indicate that the metabolic effects of GDM on offspring begin as early as infancy and can persist into adulthood [91-94]. However, how signals are transmitted across the placenta to offspring and induce epigenetic changes remains unclear.
In this context, exosomes have attracted growing attention as molecular carriers capable of transporting maternal bioactive cargos to the fetus, thereby modulating epigenetic and signaling pathways and exerting more profound and long-lasting effects on offspring metabolism [56, 95, 96]. Given the selective recognition and internalization of exosomes by placental trophoblast cells, the pathogenic exosomes in GDM mothers can directly deliver the abnormal molecular signals to the fetus, influencing the growth, development, and metabolism of the offspring [17, 22, 97, 98]. On the other hand, upon sensing abnormal maternal metabolic cues the placenta itself also secretes exosomes into the fetal circulation, thereby amplifying or transducing the maternal metabolic stress to the developing fetus [43, 99, 100]. In this section, we further explored the role of exosomes in regulating offspring metabolism during maternal-fetal communication, organized by their actions on different target organs and cell types (Figure 3).
The mechanism of GDM exosomes affecting offspring metabolism. Maternal exosomes in GDM exert multiple effects on the offspring metabolism. These exosomes enter the fetus through the placenta and then disrupt placental function via trophoblast cells, regulate fetal growth through umbilical vein endothelial cells, and modulate glucose and lipid metabolism by impacting the pancreas, adipose tissue, and liver. Abbreviations: AT-exos: Adipose Tissue-derived Exosomes; CEBPα: CCAAT/Enhancer Binding Protein α; DVL3: Dishevelled Segment Polarity Protein 3; Erk1/2: Extracellular Signal-Regulated Kinases 1 and 2; FABP4: Fatty Acid Binding Protein 4; FAS: Fatty Acid Synthase; FoxO1: Forkhead Box Protein O1; GDM: Gestational Diabetes Mellitus; GLP-1/GLP-1R: Glucagon-like Peptide-1 / GLP-1 Receptor; HSP90AA1: Heat Shock Protein 90 Alpha Family Class A Member 1; ICAM-1: Intercellular Adhesion Molecule 1; IGF pathway: Insulin-like Growth Factor pathway; MEOX2: Mesenchyme Homeobox 2; mTORC1: Mechanistic Target of Rapamycin Complex 1; NOX4: NADPH Oxidase 4; PCYOX1: Prenylcysteine Oxidase 1; PGC-1α: Peroxisome Proliferator-activated Receptor Gamma Coactivator 1-α; PI3K: Phosphoinositide 3-Kinase; PIK3R2: Phosphoinositide-3-Kinase Regulatory Subunit 2; Plin1: Perilipin 1; PPARγ: Peroxisome Proliferator-activated Receptor γ; PYGM: Muscle Glycogen Phosphorylase; SREBP-1: Sterol Regulatory Element-binding Protein 1; UVECs: Umbilical Vein Endothelial Cells; VEGF: Vascular Endothelial Growth Factor; Wnt/β-catenin: Wingless-related Integration site /β-catenin.
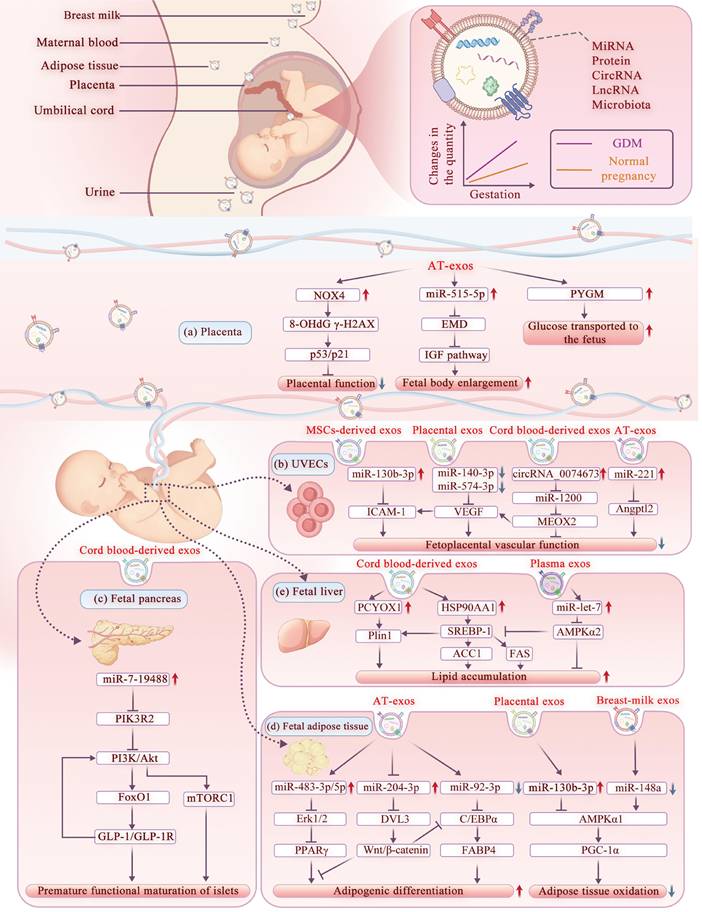
4.1 Targeting the placenta
The placenta is a critical interface between maternal and fetal circulation, facilitating molecular exchanges via exosomes [101, 102]. In metabolic diseases, normal placental development is disrupted, increasing the risk of adverse pregnancy outcomes [31, 103]. Exosomes from GDM mothers can directly act on placental trophoblasts, regulating the gene expression of oxidative stress [98], glucose, and lipid transport proteins [104], thus affecting fetal growth and development [105].
Specifically, the adipose tissue of obese mothers secretes exosomes rich in NADPH oxidase 4 (NOX4), which can be transported to trophoblast cells, upregulate the levels of 8-OHdG and γ-H2AX, activate the P53/P21 axis, resulting in severe oxidative DNA damage, premature placental aging, and adverse pregnancy outcomes [98]. Quantitatively, GDM adipose exosomes are 1.7 times more abundant than in healthy mothers and correlate positively with birth weight Z-score [104]. This effect is linked to GDM exosomes boosting glycolysis genes (PGK2, GCK) and suppressing mitochondrial gene SUCLA2 in placental cells, indicating that the glycolytic pathway was enhanced and mitochondrial function was impaired [104, 106, 107]. The increase in PYGM involved in glycogenolysis suggests an increased transfer of glucose to the fetus, which may lead to fetal overgrowth [104, 108]. Similarly, injection of adipose tissue-derived EVs loaded with miR-515-5p into pregnant mice led to increased fetal body weight and blood glucose levels [105]. miR-515-5p may enhance placental glucose uptake by targeting the EMD gene [105], which encodes the Emerin protein that regulates the IGF signaling pathway [109]. These studies have demonstrated the potential role of GDM exosomes in regulating placental function, fetal growth, and metabolism.
4.2 Targeting the umbilical vein endothelial cells (UVECs)
In the placental villous structure, UVECs line the luminal interface of the fetal side circulation [110]. As the core unit for material exchange between the fetus and the mother, its function and transported blood components reflect the fetal metabolic status to some degree [111]. Primary HUVECs obtained from GDM women exhibited persistent inflammatory and oxidative phenotypes under normal glucose levels, and this “glucose memory effect” could increase metabolic disease risk in offspring through epigenetics [112, 113]. In this process, the exosomes of GDM mothers can directly act on the UVECs. By modulating UVEC proliferation and migration, these exosomal signals may disrupt fetoplacental vascular function, thereby affecting fetal growth and development.
For instance, levels of miR-140-3p and miR-574-3p were reduced in placenta tissue-derived exosomes from GDM dams. This reduction offsets the inhibition of VEGF expression, resulting in abnormal proliferation, migration, and tube formation of UVECs, which may ultimately impair fetal development [114]. Furthermore, Gao Z et al. found that miR-130b-3p was elevated in human placental mesenchymal stem cells (MSCs)-derived exosomes, targeting ICAM-1 downregulation and inhibiting UVEC proliferation and angiogenesis [19]. Furthermore, circRNA_0074673 was upregulated in umbilical cord blood exosomes from GDM mothers, where it sponged miR-1200 to upregulate MEOX2 [115], which is associated with endothelial cell aging and migration [116], inhibiting endothelial cell migration and angiogenesis. Additionally, miR-221, which is enriched in exosomes from obese mice, inhibits endothelial cell proliferation and angiogenesis by downregulating Angptl2 expression via its 3' untranslated region, contributing to placental dysplasia during pregnancy, which impacts fetal development [117]. However, current studies have primarily documented the immediate effects of these exosomes on UVECs. Their long-term consequences for offspring's health warrant further investigation.
4.3 Targeting the fetal pancreas
The human pancreas develops from the endoderm during early embryogenesis through the formation of pancreatic buds, branching morphogenesis, and cell differentiation, establishing a primitive framework with both endocrine and exocrine functions by late gestation [118, 119]. However, critical regulatory mechanisms such as precise control of insulin secretion remain incomplete until after birth [120]. This intricate process can be disrupted by maternal hyperglycemia, interfering with pancreatic development. Studies indicate significant changes in pancreatic diameter, pancreatic mass, and β-cell mass in the offspring of HFD dams after birth [121, 122]. In fact, pancreatic remodeling may have occurred as early as the fetal period, and exosomes may play an important role in this process.
Zou L et al. demonstrated that exosomes administered to pregnant mice can cross the placenta to reach the fetal pancreas [22]. Exosomes derived from umbilical cord blood of GDM mothers were shown to enhance the expression of GLP-1R, p-Akt, and p-S6 in fetal islet β cells of mice, while also increasing the expression of disallowed genes [22]. This induces a state of precocious maturation in the fetal pancreas before birth. They further injected exosomes from the cord blood of GDM mothers into pregnant rats [46]. These exosomes, which were enriched with miR-7-19488, targeted PIK3R2 mRNA in the pancreatic islets of the fetal rats [46], leading to p85β translation arrest and subsequent activation of PI3K to phosphorylate Akt [46]. The PI3K/Akt pathway serves as a critical regulator of fetal growth [123]. Its activation enhances FoxO1 and mTORC1 signaling pathways, promoting the development and maturation of α and β cells. This process also intensifies stimulation of the local GLP-1/GLP-1R axis [46]. Consequently, these events drive the premature functional maturation of pancreatic islets during early prenatal stages, predisposing the offspring to metabolic diseases in later life [22].
4.4 Targeting the fetal adipose tissue
The body composition of the fetus may be determined as early as the first trimester when the gastrula forms [124]. During the mid-trimester of pregnancy, adipogenesis begins. MSCs first develop into preadipocytes, which then proliferate and differentiate into adipocytes in the late trimester [125]. This process will continue in several years after birth through hyperplasia and hypertrophy of adipocytes, which is highly plastic and can be programmed by the intrauterine environment, influencing the long-term metabolic health of offspring [126].
Maternal exosomes significantly influence fetal adipocyte differentiation, thereby modulating fetal growth and metabolic programming. This is evidenced by the differential expression of specific miRNAs in association with neonatal adiposity [127]. For instance, plasma exosomal miR-483-3p is significantly elevated in mothers of obese neonates compared to those of lean neonates [127], which is associated with lipid accumulation and adipocyte differentiation in low birth weight adults [128], suggesting a persistent effect. Moreover, the level of miR-483-5p is markedly increased in the umbilical cord blood exosomes of obese neonates [127]. This miRNA promotes adipogenesis in 3T3-L1 preadipocytes by suppressing the Erk1/2 signaling pathway [129], thereby increasing the risk of long-term metabolic disorders. Other miRNAs, including has-miR-204-3p and has-miR-15a, are also significantly altered in the cord blood exosomes of obese neonates [130, 131]. These miRNAs have been demonstrated to regulate adipose differentiation and intramuscular fat synthesis [130, 131]. Notably, has-miR-92a-3p and has-miR-181, which are associated with adipose differentiation and brown adipose tissue activity, are differentially expressed in umbilical cord blood of obese neonates, suggesting their potential role in the development of fetal adipose tissue [127].
Maternal exosomes play a significant role in modulating lipid synthesis and degradation in offspring. Studies have revealed an inverse correlation between birth weight and the expression levels of specific miRNAs [132]. For instance, the downregulation of miR-130b-3p, miR-29a-3p, and miR-let-7a-5p in the placenta, are all associated with increased birth weight, suggesting a potential role for these miRNAs in regulating lipid metabolism of offspring [132]. Jiang et al. demonstrated that high glucose conditions promote the secretion of exosomes enriched with miR-130b-3p from placental trophoblast cells. These exosomes suppress the expression of energy metabolism proteins, including AMPKα1 and PGC-1α [133], which is linked to impaired adipose tissue oxidation in the fetus [113]. This metabolic disruption, if sustained, can easily lead to metabolic abnormalities in the offspring [113]. Moreover, miRNA levels in breast milk exosomes have been correlated with body composition in infants. At 1 month of age, the abundance of miR-148a and miR-30b in breast milk exosomes was lowered in the infants born to obese mothers compared to controls [134]. Reduced miR-148a levels may disrupt adipogenesis and insulin signaling by targeting AMPKα1, IGF-1, and the PI3K/AKT pathway, leading to increased fat mass in infants one month after birth [134]. In contrast, miR-30b was positively correlated with infant weight and fat mass, with its overexpression stimulating lipogenesis and increasing lipid droplet size [134]. Given the relatively immature gastrointestinal tract of newborns, efficient molecular communication between mother and infant via exosomal miRNAs may regulate infant growth and fat mass.
4.5 Targeting the fetal liver
During the fetal period, the liver primarily functions in hematopoiesis, and its metabolic development begins to activate rather than being activated only after birth [135]. Recent studies have revealed that maternal high-fat diet can alter the expression of key genes involved in lipid and glucose metabolism in the fetal liver before birth through epigenetic mechanisms, leading to intrahepatic lipid accumulation [136-138]. This early programmed alteration may be partially mediated by exosome-driven signaling, ultimately impairing the metabolic adaptation and reprogramming capacity in the liver of offspring when faced with postnatal nutritional transitions [139].
Díaz M et al., found that the upregulation of both PCYOX1 and HSP90AA1 in exosomes derived from the umbilical cord blood of small-for-gestational-age (SGA) infants can predict hepatic adipose accumulation by the age of seven [45]. Mechanistically, PCYOX1 is involved in degrading prenylated proteins, and PCYOX1-deficient mice on a high-fat diet exhibit reduced body weight and visceral fat [140]. Similarly, HSP90AA1, a highly conserved molecular chaperone, promotes hepatic lipid accumulation in mice by modulating the activity of SREBP-1 [141]. Furthermore, let-7c-5p level was significantly reduced in plasma exosomes of GDM mothers during early pregnancy [49]. Let-7c-5p regulates glucose metabolism by targeting IRS1, IRS2, forkhead box protein 1 (FOXO1), and PPAR [132]. In the offspring of obese dams, increased miR-let-7 levels downregulated AMPKα2 in the liver, thereby disrupting early hepatic metabolism [142]. However, due to the limited research on the role of exosomes in regulating fetal liver metabolic development, more studies are needed to further clarify the potential mechanism.
In summary, maternal exosomes in GDM exert multiple effects on the offspring. They disrupt placental function by targeting trophoblast cells, regulate fetal growth and development by acting on umbilical vein endothelial cells, and modulate glucose and lipid metabolism by influencing the development and function of the pancreas, adipose tissue, and liver. However, these studies primarily focus on the immediate effects of exosomes on the fetus, while their long-term metabolic impacts require further investigation. Additionally, these exosomes may influence fetal immune system programming, which in turn could affect offspring metabolism. Nevertheless, the role of exosomes in this specific process is still poorly understood. Unraveling these developmental dysregulations is critical, as they may have implications for metabolic health in the offspring, warranting in-depth investigation in future studies.
5. Application of Exosomes in Reducing Metabolic Diseases in Offspring
The evidence reviewed above demonstrates that maternal-derived exosomes exert programming effects on offspring metabolism. These mechanistic insights not only establish exosomes as key signaling vectors in mediating either pathogenic or protective effects, but also provide a robust biological rationale for their application in the early prediction and intervention of metabolic diseases in the offspring.
5.1 Early prediction using exosomal biomarkers
Changes in exosomal cargo mirror early molecular events in GDM and fetal metabolic programming, often preceding overt clinical phenotypes, which makes them attractive minimally invasive biomarkers [43]. For example, plasma exosome hsa_cirRNA_0039480 is highly expressed in GDM patients at different stages, supporting its utility as an early detection biomarker [61]. Early-pregnancy placenta-derived exosomes hsa-miR-3665 and hsa-miR-6727-5p levels varied with pancreas beta cell function in GDM, which have been implicated in placental dysfunction [143]. Ye et al. further demonstrated that a panel of five plasma-derived exosomal miRNAs achieved an AUC of 0.82 for early GDM prediction [65]. These examples illustrate how exosomal profiling can capture molecular perturbations before clinical manifestations appear, enabling earlier diagnosis and intervention.
5.2 Engineered exosomes and delivery strategies
Beyond diagnostics, exosomes are increasingly transitioning from passive biomarkers to active interventional tools. However, current studies largely focus on adult metabolic disease models, and the translational gap can be bridged by engineering strategies grounded in the intergenerational communication pathways reviewed above.
One straightforward approach is harnessing native exosomes from healthy sources. Based on the evidence that maternal exosomes from normal pregnancies actively improve offspring metabolic outcomes, several studies have administered placenta- or umbilical cord-derived exosomes from healthy donors to GDM mouse models. This approach has been shown to enhance maternal glucose tolerance and insulin sensitivity [22], and to ameliorate insulin resistance and obesity in the offspring [46, 144]. Beyond these pregnancy-specific sources, stem cells represent another promising reservoir of therapeutic exosomes. For example, ADSC-derived exosomes promote M2 macrophage polarization and reduce white adipose tissue mass in obesity models [145]. These studies suggest that replenishing “protective” exosomal signals may counteract the maladaptive programming induced by adverse intrauterine environments.
Given the key role of specific miRNAs and proteins in mediating maternal-offspring metabolic crosstalk, engineering exosomes to enrich these bioactive molecules is a rational step. Engineered exosomes have been explored in various conditions, including diabetes [146], obesity [147], and aging-related disorders [148]. In the context of GDM, some exosomal miRNAs are significantly dysregulated, including miR-122-5p, miR-92a-3p, miR-29, etc. Given their potential to cross the placental barrier and influence fetal development, maternal administration of engineered exosomes loaded with these specific miRNAs may modulate offspring metabolism, an approach supported by emerging experimental evidence. For instance, injection of adipose tissue-derived EVs loaded with miR-515-5p into pregnant mice led to increased fetal body weight and blood glucose levels [105]. Together, these findings support the feasibility of designing exosomes that carry defined miRNA payloads to intervene in specific pathways during critical developmental windows in offspring.
Beyond cargo modulation, engineering the surface of exosomes for targeted delivery is a forward-looking strategy. The natural tropism of placental exosomes for fetal organs offers a blueprint for targeted delivery [149]. Future strategies may involve surface modification (e.g., with tissue-specific homing peptides) to enhance accumulation in offspring liver, pancreas, or hypothalamus [150, 151]. Alternatively, hybrid nanovesicles that combine exosomal membrane proteins with synthetic carriers could offer a more scalable and biocompatible option [152, 153].
Collectively, these findings suggest that if substances (e.g., miRNA, proteins) that have significant changes in GDM or offspring can be constructed into engineered exosomes and applied in clinical therapy, this may provide new ideas for the prevention and treatment of GDM and offspring metabolic diseases [154].
6. Conclusion
In this review, we summarize—across miRNAs, proteins, other non-coding RNAs, and microbiome-related cargos—the characteristic changes of GDM-associated maternal exosomes in blood, placenta, adipose tissue and other sources, and relate them to their actions on the placenta, fetal metabolic organs, and long-term offspring phenotypes. To our knowledge, this is the first review to systematically connect maternal exosomal alterations in GDM with organ-targeted effects in the offspring, thereby providing an integrated view of how exosomes may transmit maternal metabolic stress across generations (Figure 4).
The effects and applications of exosomes on offspring metabolism. Exosomes from GDM mothers cross the placenta into the fetus, affecting placental function and the metabolic development of umbilical vein endothelial cells, fetal pancreas, adipose tissue, and liver through epigenetic mechanisms, thereby affecting the metabolic health of offspring in both short-term and long-term perspectives. MiRNAs in exosomes can serve as early diagnostic markers for GDM, and engineered exosome interventions may help reduce the incidence of metabolic diseases in the future.
At the same time, our synthesis also reveals important limitations in the current literature. First, there is a striking lack of full “end-to-end” causal evidence that directly links: (i) the GDM state; (ii) defined changes in maternal exosomal cargo; (iii) exosome trafficking across the placenta and uptake by specific fetal organs; (iv) persistent metabolic alterations in the offspring. Most studies examine only one or two segments of this chain. Second, the cellular origins and trafficking routes of exosomes within the maternal-fetal interface remain incompletely defined. Although placenta- and cord blood-derived exosomes are frequently studied, it is often unclear whether they are of maternal, fetal, or placental origin. Third, most data remain correlative and short-term, focusing on exosomal signatures in pregnancy or early life, with relatively few studies linking maternal exosome profiles to metabolic outcomes in adolescence or adulthood. Finally, translation toward intervention is still preliminary: while engineered or “healthy pregnancy-derived” exosomes show promise in preclinical models, offspring-targeted delivery and safety, as well as standardized production and characterization, remain challenging.
Future research should therefore prioritize multi-level, longitudinal, and mechanistic designs. In humans, prospective pregnancy cohorts integrating maternal metabolic phenotyping with serial profiling of exosomal cargo and long-term follow-up of offspring metabolic outcomes will be essential to map temporal trajectories and identify robust, early biomarkers of risk. In parallel, causal studies in animal models are needed to manipulate exosomes along the entire maternal-placental-fetal axis—for example, by selectively altering exosome release in maternal adipose tissue or placenta, genetically or pharmacologically editing key exosomal cargos, or transferring well-defined exosome populations from GDM versus control mothers into naïve pregnancies, and then tracking offspring organ-specific metabolism. Advancing exosome-tracing technologies (e.g., genetic reporters, in vivo imaging, placental-on-a-chip and organoid models) will be critical to visualize exosome trafficking across the placental barrier and their uptake by fetal liver, pancreas, adipose tissue, skeletal muscle, and brain.
The clinical translation of engineered exosomes is accelerating, marked by the emergence of clinical studies that have reported preliminary safety and efficacy [155, 156]. However, the application of these strategies in metabolic diseases remains immature. On the translational side, the therapeutic potential of exosomes in preventing offspring metabolic disease will depend on transitioning from observational administration to mechanism-based design. These mechanistic insights we reviewed above can be translated into engineering strategies: parental cell engineering to overexpress protective miRNAs implicated in metabolic programming; post-isolation loading of small molecules or siRNAs targeting key metabolic regulators; and exosome-inspired synthetic vesicles that replicate the functional properties of placental or milk exosomes while enabling production.
Future work should also focus on optimizing loading strategies for exosomes, and rigorously evaluating safety, dosing, and timing of administration during critical developmental windows. Standardization of exosome isolation, characterization, and nomenclature will also be required to enable cross-study comparison and clinical implementation. If these gaps can be addressed, exosome-based strategies-combining their biomarker value (capturing molecular events before overt phenotypes) with their therapeutic carrier function-may ultimately provide a novel avenue to reduce the long-term metabolic burden of GDM for both mothers and their children, transforming early-life prevention of metabolic diseases.
Abbreviations
ACLY: ATP Citrate Lyase; ADSCs: Adipose-Derived Stem Cells; AKT: Protein Kinase B; BANF-1: Barrier To Autointegration Factor; CARHSP1: Calcium-Regulated Heat-Stable Protein 1; DNMT: DNA Methyltransferase; DOHaD: The Developmental Origins of Health and Disease; EVs: Extracellular Vesicles; GDM: Gestational Diabetes Mellitus; GW: Gestational Weeks; ICAM-1: Intercellular Adhesion Molecule-1; MTTP: Microsomal Triglyceride Transfer Protein; MVBs: Multivesicular Bodies; NAFLD: Non-Alcoholic Fatty Liver Disease; NGT: Normal Glucose Tolerance; NOX4: NADPH Oxidase 4; NTA: Nanoparticle Tracking Analysis; Sirt1: Sirtuin 1; SOCS2: Suppressor of Cytokine Signaling 2; SREBP-1c: Sterol Regulatory Element-Binding Protein-1C; Thbs1: Thrombospondin 1; UVECs: Umbilical Vein Endothelial Cells.
Acknowledgements
Funding
This work was supported by the grants from National Natural Science Foundation of China (No. 82570971, 82470873, 82170854), Capital's Funds for Health Improvement and Research (2026-1-4011), Science and Technology Research Project of China Science and Technology Development Center for Chinese Medicine (CXZH2024059), National High Level Hospital Clinical Research Funding (2025-PUMCH-C-021, 2022-PUMCH-C-019), CAMS Innovation Fund for Medical Sciences (CIFMS2021-1-I2M-002), the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2023-PT320-10), Beijing Natural Science Foundation (7202163), Beijing Municipal Science & Technology Commission (Z201100005520011).
Author Contributions
J.Z. and D.W. wrote the manuscript and drew the figures. J.Z., D.W., and J.L. collected the related papers, M.Y., X.Z., and Y.S., created the Tables, D.W., and J.L. helped to revise the manuscript. D.W., and J.L., and X.X. designed and revised the manuscript. All the authors read and approved the final version of the review.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB. et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes research and clinical practice. 2022;183:109119.doi 10.1016/j.diabres.2021.109119
2. Sweeting A, Wong J, Murphy HR, Ross GP. A Clinical Update on Gestational Diabetes Mellitus. Endocrine reviews. 2022;43:763-93.doi 10.1210/endrev/bnac003
3. Godfrey KM, Reynolds RM, Prescott SL, Nyirenda M, Jaddoe VW, Eriksson JG. et al. Influence of maternal obesity on the long-term health of offspring. The lancet Diabetes & endocrinology. 2017;5:53-64.doi 10.1016/s2213-8587(16)30107-3
4. Hoffman DJ, Powell TL, Barrett ES, Hardy DB. Developmental origins of metabolic diseases. Physiological reviews. 2021;101:739-95.doi 10.1152/physrev.00002.2020
5. Foo RX, Ma JJ, Du R, Goh GBB, Chong YS, Zhang C. et al. Gestational diabetes mellitus and development of intergenerational non-alcoholic fatty liver disease (NAFLD) after delivery: a systematic review and meta-analysis. EClinicalMedicine. 2024;72:102609.doi 10.1016/j.eclinm.2024.102609
6. Furse S, Koulman A, Ozanne SE, Poston L, White SL, Meek CL. Altered Lipid Metabolism in Obese Women With Gestational Diabetes and Associations With Offspring Adiposity. The Journal of clinical endocrinology and metabolism. 2022;107:e2825-e32.doi 10.1210/clinem/dgac206
7. Berglund SK, García-Valdés L, Torres-Espinola FJ, Segura MT, Martínez-Zaldívar C, Aguilar MJ. et al. Maternal, fetal and perinatal alterations associated with obesity, overweight and gestational diabetes: an observational cohort study (PREOBE). BMC public health. 2016;16:207.doi 10.1186/s12889-016-2809-3
8. Metzger BE, Lowe LP, Dyer AR, Trimble ER, Chaovarindr U, Coustan DR. et al. Hyperglycemia and adverse pregnancy outcomes. The New England journal of medicine. 2008;358:1991-2002.doi 10.1056/NEJMoa0707943
9. Meyrueix LP, Gharaibeh R, Xue J, Brouwer C, Jones C, Adair L. et al. Gestational diabetes mellitus placentas exhibit epimutations at placental development genes. Epigenetics. 2022;17:2157-77.doi 10.1080/15592294.2022.2111751
10. Alba-Linares JJ, Pérez RF, Tejedor JR, Bastante-Rodríguez D, Ponce F, Carbonell NG. et al. Maternal obesity and gestational diabetes reprogram the methylome of offspring beyond birth by inducing epigenetic signatures in metabolic and developmental pathways. Cardiovascular diabetology. 2023;22:44.doi 10.1186/s12933-023-01774-y
11. Lai JJ, Chau ZL, Chen SY, Hill JJ, Korpany KV, Liang NW. et al. Exosome Processing and Characterization Approaches for Research and Technology Development. Advanced science (Weinheim, Baden-Wurttemberg, Germany). 2022;9:e2103222.doi 10.1002/advs.202103222
12. Yang B, Chen Y, Shi J. Exosome Biochemistry and Advanced Nanotechnology for Next-Generation Theranostic Platforms. Advanced materials (Deerfield Beach, Fla). 2019;31:e1802896.doi 10.1002/adma.201802896
13. Fuchs A, Samovski D, Smith GI, Cifarelli V, Farabi SS, Yoshino J. et al. Associations Among Adipose Tissue Immunology, Inflammation, Exosomes and Insulin Sensitivity in People With Obesity and Nonalcoholic Fatty Liver Disease. Gastroenterology. 2021;161:968-81.e12.doi 10.1053/j.gastro.2021.05.008
14. Ying W, Gao H, Dos Reis FCG, Bandyopadhyay G, Ofrecio JM, Luo Z. et al. MiR-690, an exosomal-derived miRNA from M2-polarized macrophages, improves insulin sensitivity in obese mice. Cell metabolism. 2021;33:781-90.e5.doi 10.1016/j.cmet.2020.12.019
15. Wang J, Li L, Zhang Z, Zhang X, Zhu Y, Zhang C. et al. Extracellular vesicles mediate the communication of adipose tissue with brain and promote cognitive impairment associated with insulin resistance. Cell metabolism. 2022;34:1264-79.e8.doi 10.1016/j.cmet.2022.08.004
16. Salomon C, Torres MJ, Kobayashi M, Scholz-Romero K, Sobrevia L, Dobierzewska A. et al. A gestational profile of placental exosomes in maternal plasma and their effects on endothelial cell migration. PloS one. 2014;9:e98667.doi 10.1371/journal.pone.0098667
17. Salomon C, Scholz-Romero K, Sarker S, Sweeney E, Kobayashi M, Correa P. et al. Gestational Diabetes Mellitus Is Associated With Changes in the Concentration and Bioactivity of Placenta-Derived Exosomes in Maternal Circulation Across Gestation. Diabetes. 2016;65:598-609.doi 10.2337/db15-0966
18. Li H, Yang H, Liu J, Yang H, Gao X, Yang X. et al. Adipose stem cells-derived small extracellular vesicles transport Thrombospondin 1 cargo to promote insulin resistance in gestational diabetes mellitus. Diabetology & metabolic syndrome. 2024;16:105.doi 10.1186/s13098-024-01276-1
19. Gao Z, Wang N, Liu X. Human placenta mesenchymal stem cell-derived exosome shuttling microRNA-130b-3p from gestational diabetes mellitus patients targets ICAM-1 and perturbs human umbilical vein endothelial cell angiogenesis. Acta diabetologica. 2022;59:1091-107.doi 10.1007/s00592-022-01910-2
20. Sheller-Miller S, Choi K, Choi C, Menon R. Cyclic-recombinase-reporter mouse model to determine exosome communication and function during pregnancy. American journal of obstetrics and gynecology. 2019;221:502.e1-e12.doi 10.1016/j.ajog.2019.06.010
21. Klemetti MM, Pettersson ABV, Ahmad Khan A, Ermini L, Porter TR, Litvack ML. et al. Lipid profile of circulating placental extracellular vesicles during pregnancy identifies foetal growth restriction risk. J Extracell Vesicles. 2024;13:e12413.doi 10.1002/jev2.12413
22. Zou L, Xu C, Wang L, Cao X, Jia X, Yang Z. et al. Human gestational diabetes mellitus-derived exosomes impair glucose homeostasis in pregnant mice and stimulate functional maturation of offspring-islets. Life sciences. 2024;342:122514.doi 10.1016/j.lfs.2024.122514
23. Edwards PD, Lavergne SG, McCaw LK, Wijenayake S, Boonstra R, McGowan PO. et al. Maternal effects in mammals: Broadening our understanding of offspring programming. Frontiers in neuroendocrinology. 2021;62:100924.doi 10.1016/j.yfrne.2021.100924
24. Abbas MA, Al-Saigh NN, Saqallah FG. Regulation of adipogenesis by exosomal milk miRNA. Reviews in endocrine & metabolic disorders. 2023;24:297-316.doi 10.1007/s11154-023-09788-3
25. Doyle LM, Wang MZ. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019 8.doi:10.3390/cells8070727
26. van Niel G, D'Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nature reviews Molecular cell biology. 2018;19:213-28.doi 10.1038/nrm.2017.125
27. Kim HI, Park J, Zhu Y, Wang X, Han Y, Zhang D. Recent advances in extracellular vesicles for therapeutic cargo delivery. Experimental & molecular medicine. 2024;56:836-49.doi 10.1038/s12276-024-01201-6
28. Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. The Journal of cell biology. 2013;200:373-83.doi 10.1083/jcb.201211138
29. Mathieu M, Martin-Jaular L, Lavieu G, Théry C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nature cell biology. 2019;21:9-17.doi 10.1038/s41556-018-0250-9
30. Arya SB, Collie SP, Parent CA. The ins-and-outs of exosome biogenesis, secretion, and internalization. Trends in cell biology. 2024;34:90-108.doi 10.1016/j.tcb.2023.06.006
31. Sun Q, Chang H, Wang H, Zheng L, Weng Y, Zheng D. et al. Regulatory roles of extracellular vesicles in pregnancy complications. Journal of Advanced Research. 2025 doi:10.1016/j.jare.2025.02.010
32. Hoshino A, Kim HS, Bojmar L, Gyan KE, Cioffi M, Hernandez J. et al. Extracellular Vesicle and Particle Biomarkers Define Multiple Human Cancers. Cell. 2020;182:1044-61.e18.doi 10.1016/j.cell.2020.07.009
33. Liu X, Teng X, Wang J, Zhang G, Guo W, Zhang W. et al. Exosome-based mucosal therapeutic and diagnostic system: towards clinical translation. Journal of controlled release: official journal of the Controlled Release Society. 2025;385:113966.doi 10.1016/j.jconrel.2025.113966
34. Lucien F, Gustafson D, Lenassi M, Li B, Teske JJ, Boilard E. et al. MIBlood-EV: Minimal information to enhance the quality and reproducibility of blood extracellular vesicle research. Journal of extracellular vesicles. 2023;12:e12385.doi 10.1002/jev2.12385
35. Welsh JA, Goberdhan DCI, O'Driscoll L, Buzas EI, Blenkiron C, Bussolati B. et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. Journal of extracellular vesicles. 2024;13:e12404.doi 10.1002/jev2.12404
36. Dad HA, Gu TW, Zhu AQ, Huang LQ, Peng LH. Plant Exosome-like Nanovesicles: Emerging Therapeutics and Drug Delivery Nanoplatforms. Molecular therapy: the journal of the American Society of Gene Therapy. 2021;29:13-31.doi 10.1016/j.ymthe.2020.11.030
37. Ye J, Li D, Jie Y, Luo H, Zhang W, Qiu C. Exosome-based nanoparticles and cancer immunotherapy. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2024;179:117296.doi 10.1016/j.biopha.2024.117296
38. Sabapatha A, Gercel-Taylor C, Taylor DD. Specific isolation of placenta-derived exosomes from the circulation of pregnant women and their immunoregulatory consequences. American journal of reproductive immunology (New York, NY: 1989). 2006;56:345-55.doi 10.1111/j.1600-0897.2006.00435.x
39. Sun Q, Chang H, Wang H, Zheng L, Weng Y, Zheng D. et al. Regulatory roles of extracellular vesicles in pregnancy complications. J Adv Res. 2025 doi:10.1016/j.jare.2025.02.010
40. Nair S, Salomon C. Extracellular vesicles as critical mediators of maternal-fetal communication during pregnancy and their potential role in maternal metabolism. Placenta. 2020;98:60-8.doi 10.1016/j.placenta.2020.06.011
41. Jin J, Menon R. Placental exosomes: A proxy to understand pregnancy complications. American journal of reproductive immunology (New York, NY: 1989). 2018;79:e12788.doi 10.1111/aji.12788
42. Czernek L, Düchler M. Exosomes as Messengers Between Mother and Fetus in Pregnancy. International journal of molecular sciences. 2020 21.doi:10.3390/ijms21124264
43. Rosenfeld CS. Placenta Extracellular Vesicles: Messengers Connecting Maternal and Fetal Systems. Biomolecules. 2024 14.doi:10.3390/biom14080995
44. Paul N, Sultana Z, Fisher JJ, Maiti K, Smith R. Extracellular vesicles- crucial players in human pregnancy. Placenta. 2023;140:30-8.doi 10.1016/j.placenta.2023.07.006
45. Díaz M, Quesada-López T, Villarroya F, Casano P, López-Bermejo A, de Zegher F. et al. The Proteome of Exosomes at Birth Predicts Insulin Resistance, Adrenarche and Liver Fat in Childhood. International journal of molecular sciences. 2025 26.doi:10.3390/ijms26041721
46. Xu C, Zou L, Wang L, Lv W, Cao X, Jia X. et al. Gestational diabetes mellitus-derived miR-7-19488 targets PIK3R2 mRNA to stimulate the abnormal development and maturation of offspring-islets. Life sciences. 2025;363:123369.doi 10.1016/j.lfs.2025.123369
47. Kunte P, Barberio M, Tiwari P, Sukla K, Harmon B, Epstein S. et al. Neonatal adiposity is associated with microRNAs in adipocyte-derived extracellular vesicles in maternal and cord blood, a discovery analysis. International journal of obesity (2005). 2024;48:403-13.doi 10.1038/s41366-023-01432-z
48. Dutta S, Lai A, Scholz-Romero K, Shiddiky MJA, Yamauchi Y, Mishra JS. et al. Hypoxia-induced small extracellular vesicle proteins regulate proinflammatory cytokines and systemic blood pressure in pregnant rats. Clinical science (London, England: 1979). 2020;134:593-607.doi 10.1042/cs20191155
49. James-Allan LB, Devaskar SU. Extracellular vesicles and their role in gestational diabetes mellitus. Placenta. 2021;113:15-22.doi 10.1016/j.placenta.2021.02.012
50. Jayabalan N, Lai A, Ormazabal V, Adam S, Guanzon D, Palma C. et al. Adipose Tissue Exosomal Proteomic Profile Reveals a Role on Placenta Glucose Metabolism in Gestational Diabetes Mellitus. The Journal of clinical endocrinology and metabolism. 2019;104:1735-52.doi 10.1210/jc.2018-01599
51. Liu T, Sun YC, Cheng P, Shao HG. Adipose tissue macrophage-derived exosomal miR-29a regulates obesity-associated insulin resistance. Biochemical and biophysical research communications. 2019;515:352-8.doi 10.1016/j.bbrc.2019.05.113
52. Hijmans JG, Bammert TD, Stockelman KA, Reiakvam WR, Greiner JJ, DeSouza CA. High glucose-induced endothelial microparticles increase adhesion molecule expression on endothelial cells. Diabetology international. 2019;10:143-7.doi 10.1007/s13340-018-0375-x
53. Fang X, Zhang Y, Zhang Y, Guan H, Huang X, Miao R. et al. Endothelial extracellular vesicles: their possible function and clinical significance in diabetic vascular complications. Journal of translational medicine. 2024;22:944.doi 10.1186/s12967-024-05760-0
54. James-Allan LB, Rosario FJ, Barner K, Lai A, Guanzon D, McIntyre HD. et al. Regulation of glucose homeostasis by small extracellular vesicles in normal pregnancy and in gestational diabetes. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2020;34:5724-39.doi 10.1096/fj.201902522RR
55. Seedat F, Kandzija N, Ellis MJ, Jiang S, Sarbalina A, Bancroft J. et al. Placental small extracellular vesicles from normal pregnancy and gestational diabetes increase insulin gene transcription and content in β cells. Clinical science (London, England: 1979). 2024;138:1481-502.doi 10.1042/cs20241782
56. Chen P, Gu M, Wan S, Jiang X, Zhang F, Li Y. et al. Gestational Diabetes Mellitus Impedes Fetal Lung Development Through Exosome-Dependent Crosstalk Between Trophoblasts and Lung Epithelial Cells. International journal of nanomedicine. 2023;18:641-57.doi 10.2147/ijn.S396194
57. Lin J, Zhao D, Liang Y, Liang Z, Wang M, Tang X. et al. Proteomic analysis of plasma total exosomes and placenta-derived exosomes in patients with gestational diabetes mellitus in the first and second trimesters. BMC Pregnancy Childbirth. 2024;24:713.doi 10.1186/s12884-024-06919-9
58. Nair S, Jayabalan N, Guanzon D, Palma C, Scholz-Romero K, Elfeky O. et al. Human placental exosomes in gestational diabetes mellitus carry a specific set of miRNAs associated with skeletal muscle insulin sensitivity. Clinical science (London, England: 1979). 2018;132:2451-67.doi 10.1042/cs20180487
59. Gillet V, Ouellet A, Stepanov Y, Rodosthenous RS, Croft EK, Brennan K. et al. miRNA Profiles in Extracellular Vesicles From Serum Early in Pregnancies Complicated by Gestational Diabetes Mellitus. The Journal of clinical endocrinology and metabolism. 2019;104:5157-69.doi 10.1210/jc.2018-02693
60. Shen Y, Zhang M, Zhang Z, Li M, Chen X, Zhu W. et al. Exosome miRNA Profiles: The Reduced Expression of miRNA-27a-5p and Predictive miRNAs for Gestational Diabetes Mellitus. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2025;39:e70710.doi 10.1096/fj.202500470RR
61. Jiang B, Zhang J, Sun X, Yang C, Cheng G, Xu M. et al. Circulating exosomal hsa_circRNA_0039480 is highly expressed in gestational diabetes mellitus and may be served as a biomarker for early diagnosis of GDM. Journal of translational medicine. 2022;20:5.doi 10.1186/s12967-021-03195-5
62. Filardi T, Catanzaro G, Grieco GE, Splendiani E, Trocchianesi S, Santangelo C. et al. Identification and Validation of miR-222-3p and miR-409-3p as Plasma Biomarkers in Gestational Diabetes Mellitus Sharing Validated Target Genes Involved in Metabolic Homeostasis. International journal of molecular sciences. 2022 23.doi:10.3390/ijms23084276
63. Nair S, Guanzon D, Jayabalan N, Lai A, Scholz-Romero K, Kalita de Croft P. et al. Extracellular vesicle-associated miRNAs are an adaptive response to gestational diabetes mellitus. Journal of translational medicine. 2021;19:360.doi 10.1186/s12967-021-02999-9
64. Wang Y, Yuan Y, Shen S, Ge Z, Zhu D, Bi Y. Placenta-derived exosomes exacerbate beta cell dysfunction in gestational diabetes mellitus through delivery of miR-320b. Frontiers in endocrinology. 2023;14:1282075.doi 10.3389/fendo.2023.1282075
65. Ye Z, Wang S, Huang X, Chen P, Deng L, Li S. et al. Plasma Exosomal miRNAs Associated With Metabolism as Early Predictor of Gestational Diabetes Mellitus. Diabetes. 2022;71:2272-83.doi 10.2337/db21-0909
66. Tobaruela-Resola AL, Milagro FI, Elorz M, Benito-Boillos A, Herrero JI, Mogna-Peláez P. et al. Circulating miR-122-5p, miR-151a-3p, miR-126-5p and miR-21-5p as potential predictive biomarkers for Metabolic Dysfunction-Associated Steatotic Liver Disease assessment. Journal of physiology and biochemistry. 2024 doi:10.1007/s13105-024-01037-8
67. Lee HM, Wong WKK, Fan B, Lau ES, Hou Y, O CK. et al. Detection of increased serum miR-122-5p and miR-455-3p levels before the clinical diagnosis of liver cancer in people with type 2 diabetes. Scientific reports. 2021;11:23756.doi 10.1038/s41598-021-03222-x
68. Thamotharan S, Ghosh S, James-Allan L, Lei MYY, Janzen C, Devaskar SU. Circulating extracellular vesicles exhibit a differential miRNA profile in gestational diabetes mellitus pregnancies. PloS one. 2022;17:e0267564.doi 10.1371/journal.pone.0267564
69. Willeit P, Skroblin P, Moschen AR, Yin X, Kaudewitz D, Zampetaki A. et al. Circulating MicroRNA-122 Is Associated With the Risk of New-Onset Metabolic Syndrome and Type 2 Diabetes. Diabetes. 2017;66:347-57.doi 10.2337/db16-0731
70. Du X, Li Q, Wu Z, Xue L. miR-92a-3p and miR-182-3p as potential biomarkers for the differential diagnosis of gestational diabetes mellitus and its correlation with pregnancy outcomes. Irish journal of medical science. 2025;194:909-16.doi 10.1007/s11845-025-03953-0
71. Wang W, Wang J, Yan M, Jiang J, Bian A. MiRNA-92a protects pancreatic B-cell function by targeting KLF2 in diabetes mellitus. Biochemical and biophysical research communications. 2018;500:577-82.doi 10.1016/j.bbrc.2018.04.097
72. Li S, Yuan L, Su L, Lian Z, Liu C, Zhang F. et al. Decreased miR-92a-3p expression potentially mediates the pro-angiogenic effects of oxidative stress-activated endothelial cell-derived exosomes by targeting tissue factor. International journal of molecular medicine. 2020;46:1886-98.doi 10.3892/ijmm.2020.4713
73. Li J, Chen L, Qiuqin T, Wu W, Hao G, Lou L. et al. The role, mechanism and potentially novel biomarker of microRNA-17-92 cluster in macrosomia. Scientific reports. 2015;5:17212.doi 10.1038/srep17212
74. Li J, Zhang Y, Ye Y, Li D, Liu Y, Lee E. et al. Pancreatic β cells control glucose homeostasis via the secretion of exosomal miR-29 family. Journal of extracellular vesicles. 2021;10:e12055.doi 10.1002/jev2.12055
75. Kurtz CL, Fannin EE, Toth CL, Pearson DS, Vickers KC, Sethupathy P. Inhibition of miR-29 has a significant lipid-lowering benefit through suppression of lipogenic programs in liver. Scientific reports. 2015;5:12911.doi 10.1038/srep12911
76. Chen Y, Zhu Q, Cheng L, Wang Y, Li M, Yang Q. et al. Exosome detection via the ultrafast-isolation system: EXODUS. Nature methods. 2021;18:212-8.doi 10.1038/s41592-020-01034-x
77. Yu J, Ren J, Ren Y, Wu Y, Zeng Y, Zhang Q. et al. Using metabolomics and proteomics to identify the potential urine biomarkers for prediction and diagnosis of gestational diabetes. EBioMedicine. 2024;101:105008.doi 10.1016/j.ebiom.2024.105008
78. Herrera-Van Oostdam AS, Toro-Ortíz JC, López JA, Noyola DE, García-López DA, Durán-Figueroa NV. et al. Placental exosomes isolated from urine of patients with gestational diabetes exhibit a differential profile expression of microRNAs across gestation. International journal of molecular medicine. 2020;46:546-60.doi 10.3892/ijmm.2020.4626
79. Bernea EG, Suica VI, Uyy E, Cerveanu-Hogas A, Boteanu RM, Ivan L. et al. Exosome Proteomics Reveals the Deregulation of Coagulation, Complement and Lipid Metabolism Proteins in Gestational Diabetes Mellitus. Molecules (Basel, Switzerland). 2022 27.doi:10.3390/molecules27175502
80. Musa E, Salazar-Petres E, Vatish M, Levitt N, Sferruzzi-Perri AN, Matjila MJ. Kisspeptin signalling and its correlation with placental ultrastructure and clinical outcomes in pregnant South African women with obesity and gestational diabetes. Placenta. 2024;154:49-59.doi 10.1016/j.placenta.2024.05.138
81. Lin J, Zhao D, Liang Y, Liang Z, Wang M, Tang X. et al. Proteomic analysis of plasma total exosomes and placenta-derived exosomes in patients with gestational diabetes mellitus in the first and second trimesters. BMC Pregnancy and Childbirth. 2024;24:713.doi 10.1186/s12884-024-06919-9
82. Schmeidler-Sapiro KT, Ratnoff OD, Gordon EM. Mitogenic effects of coagulation factor XII and factor XIIa on HepG2 cells. Proceedings of the National Academy of Sciences of the United States of America. 1991;88:4382-5.doi 10.1073/pnas.88.10.4382
83. Kandzija N, Payne S, Cooke WR, Seedat F, Fischer R, Vatish M. Protein Profiling of Placental Extracellular Vesicles in Gestational Diabetes Mellitus. International journal of molecular sciences. 2024 25.doi:10.3390/ijms25041947
84. Jiang Y, Wang Y, Xue K, Ma J, Xu S, Wang K. et al. The RNA binding protein CARHSP1 facilitates tumor growth, metastasis and immune escape by enhancing IL-17RA mRNA stabilization in prostate cancer. Cell & bioscience. 2025;15:33.doi 10.1186/s13578-025-01371-4
85. Yuan Y, Li Y, Hu L, Wen J. Exosomal RNA Expression Profiles and Their Prediction Performance in Patients With Gestational Diabetes Mellitus and Macrosomia. Frontiers in endocrinology. 2022;13:864971.doi 10.3389/fendo.2022.864971
86. Ma B, Wang S, Wu W, Shan P, Chen Y, Meng J. et al. Mechanisms of circRNA/lncRNA-miRNA interactions and applications in disease and drug research. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2023;162:114672.doi 10.1016/j.biopha.2023.114672
87. Dias S, Pheiffer C, Adam S. The Maternal Microbiome and Gestational Diabetes Mellitus: Cause and Effect. Microorganisms. 2023 11.doi:10.3390/microorganisms11092217
88. Wang S, Liu Y, Tam WH, Ching JYL, Xu W, Yan S. et al. Maternal gestational diabetes mellitus associates with altered gut microbiome composition and head circumference abnormalities in male offspring. Cell host & microbe. 2024;32:1192-206.e5.doi 10.1016/j.chom.2024.06.005
89. Chang CJ, Bai YC, Jiang H, Ma QW, Hsieh CH, Liu CC. et al. Microbiome analysis of serum extracellular vesicles in gestational diabetes patients. Acta diabetologica. 2025;62:329-41.doi 10.1007/s00592-024-02358-2
90. Park SY, Rao C, Coyte KZ, Kuziel GA, Zhang Y, Huang W. et al. Strain-level fitness in the gut microbiome is an emergent property of glycans and a single metabolite. Cell. 2022;185:513-29.e21.doi 10.1016/j.cell.2022.01.002
91. Keely EJ, Malcolm JC, Hadjiyannakis S, Gaboury I, Lough G, Lawson ML. Prevalence of metabolic markers of insulin resistance in offspring of gestational diabetes pregnancies. Pediatr Diabetes. 2008;9:53-9.doi 10.1111/j.1399-5448.2007.00258.x
92. Tam WH, Ma RC, Yang X, Ko GT, Tong PC, Cockram CS. et al. Glucose intolerance and cardiometabolic risk in children exposed to maternal gestational diabetes mellitus in utero. Pediatrics. 2008;122:1229-34.doi 10.1542/peds.2008-0158
93. Wright CS, Rifas-Shiman SL, Rich-Edwards JW, Taveras EM, Gillman MW, Oken E. Intrauterine exposure to gestational diabetes, child adiposity, and blood pressure. American journal of hypertension. 2009;22:215-20.doi 10.1038/ajh.2008.326
94. Gao M, Cao S, Li N, Liu J, Lyu Y, Li J. et al. Risks of overweight in the offspring of women with gestational diabetes at different developmental stages: A meta-analysis with more than half a million offspring. Obesity reviews: an official journal of the International Association for the Study of Obesity. 2022;23:e13395.doi 10.1111/obr.13395
95. Melnik BC, Stremmel W, Weiskirchen R, John SM, Schmitz G. Exosome-Derived MicroRNAs of Human Milk and Their Effects on Infant Health and Development. Biomolecules. 2021 11.doi:10.3390/biom11060851
96. Chiarello DI, Salsoso R, Toledo F, Mate A, Vázquez CM, Sobrevia L. Foetoplacental communication via extracellular vesicles in normal pregnancy and preeclampsia. Molecular aspects of medicine. 2018;60:69-80.doi 10.1016/j.mam.2017.12.002
97. Mazzarella R, Cañón-Beltrán K, Cajas YN, Hamdi M, González EM, da Silveira JC. et al. Extracellular vesicles-coupled miRNAs from oviduct and uterus modulate signaling pathways related to lipid metabolism and bovine early embryo development. Journal of animal science and biotechnology. 2024;15:51.doi 10.1186/s40104-024-01008-5
98. Tao Y, Chen W, Xu H, Xu J, Yang H, Luo X. et al. Adipocyte-Derived Exosomal NOX4-Mediated Oxidative Damage Induces Premature Placental Senescence in Obese Pregnancy. International journal of nanomedicine. 2023;18:4705-26.doi 10.2147/ijn.S419081
99. Cuman C, Van Sinderen M, Gantier MP, Rainczuk K, Sorby K, Rombauts L. et al. Human Blastocyst Secreted microRNA Regulate Endometrial Epithelial Cell Adhesion. EBioMedicine. 2015;2:1528-35.doi 10.1016/j.ebiom.2015.09.003
100. Giacomini E, Vago R, Sanchez AM, Podini P, Zarovni N, Murdica V. et al. Secretome of in vitro cultured human embryos contains extracellular vesicles that are uptaken by the maternal side. Scientific reports. 2017;7:5210.doi 10.1038/s41598-017-05549-w
101. Akhaphong B, Baumann DC, Beetch M, Lockridge AD, Jo S, Wong A. et al. Placental mTOR complex 1 regulates fetal programming of obesity and insulin resistance in mice. JCI insight. 2021 6.doi:10.1172/jci.insight.149271
102. Anam AK, Cooke KM, Dratver MB, O'Bryan JV, Perley LE, Guller SM. et al. Insulin increases placental triglyceride as a potential mechanism for fetal adiposity in maternal obesity. Molecular metabolism. 2022;64:101574.doi 10.1016/j.molmet.2022.101574
103. Nair S, Razo-Azamar M, Jayabalan N, Dalgaard LT, Palacios-González B, Sørensen A. et al. Advances in extracellular vesicles as mediators of cell-to-cell communication in pregnancy. Cytokine & growth factor reviews. 2024;76:86-98.doi 10.1016/j.cytogfr.2023.12.004
104. Jayabalan N, Lai A, Ormazabal V, Adam S, Guanzon D, Palma C. et al. Adipose Tissue Exosomal Proteomic Profile Reveals a Role on Placenta Glucose Metabolism in Gestational Diabetes Mellitus. The Journal of Clinical Endocrinology & Metabolism. 2018;104:1735-52.doi 10.1210/jc.2018-01599 %J The Journal of Clinical Endocrinology & Metabolism
105. Jayabalan N, Nair S, Lai A, Scholz-Romero K, Razo-Azamar M, Ormazabal V. et al. Extracellular vesicle-associated miR-515-5p from adipose tissue regulates placental metabolism and fetal growth in gestational diabetes mellitus. Cardiovascular diabetology. 2025;24:205.doi 10.1186/s12933-025-02739-z
106. Peng C, Jiang H, Jing L, Yang W, Guan X, Wang H. et al. Macrophage SUCLA2 coupled glutaminolysis manipulates obesity through AMPK. Nature communications. 2025;16:1738.doi 10.1038/s41467-025-57044-w
107. Wu ST, Liu B, Ai ZZ, Hong ZC, You PT, Wu HZ. et al. Esculetin Inhibits Cancer Cell Glycolysis by Binding Tumor PGK2, GPD2, and GPI. Frontiers in pharmacology. 2020;11:379.doi 10.3389/fphar.2020.00379
108. Cao M, Bai L, Wei H, Guo Y, Sun G, Sun H. et al. Dietary supplementation with pterostilbene activates the PI3K-AKT-mTOR signalling pathway to alleviate progressive oxidative stress and promote placental nutrient transport. Journal of animal science and biotechnology. 2024;15:133.doi 10.1186/s40104-024-01090-9
109. Koch AJ, Holaska JM. Emerin in health and disease. Seminars in Cell & Developmental Biology. 2014;29:95-106.doi 10.1016/j.semcdb.2013.12.008
110. Burton GJ, Jauniaux E. Development of the Human Placenta and Fetal Heart: Synergic or Independent? Frontiers in physiology. 2018;9:373.doi 10.3389/fphys.2018.00373
111. Terstappen F, Calis JJA, Paauw ND, Joles JA, van Rijn BB, Mokry M. et al. Developmental programming in human umbilical cord vein endothelial cells following fetal growth restriction. Clinical epigenetics. 2020;12:185.doi 10.1186/s13148-020-00980-9
112. Di Pietrantonio N, Sánchez-Ceinos J, Shumliakivska M, Rakow A, Mandatori D, Di Tomo P. et al. The inflammatory and oxidative phenotype of gestational diabetes is epigenetically transmitted to the offspring: role of methyltransferase MLL1-induced H3K4me3. European heart journal. 2024;45:5171-85.doi 10.1093/eurheartj/ehae688
113. Tryggestad JB, Vishwanath A, Jiang S, Mallappa A, Teague AM, Takahashi Y. et al. Influence of gestational diabetes mellitus on human umbilical vein endothelial cell miRNA. Clinical science (London, England: 1979). 2016;130:1955-67.doi 10.1042/cs20160305
114. Zhang L, Wu Q, Zhu S, Tang Y, Chen Y, Chen D. et al. Chemerin-Induced Down-Regulation of Placenta-Derived Exosomal miR-140-3p and miR-574-3p Promotes Umbilical Vein Endothelial Cells Proliferation, Migration, and Tube Formation in Gestational Diabetes Mellitus. Cells. 2022 11.doi:10.3390/cells11213457
115. Huang Y, Liang B, Chen X. Exosomal circular RNA circ_0074673 regulates the proliferation, migration, and angiogenesis of human umbilical vein endothelial cells via the microRNA-1200/MEOX2 axis. Bioengineered. 2021;12:6782-92.doi 10.1080/21655979.2021.1967077
116. Douville JM, Cheung DY, Herbert KL, Moffatt T, Wigle JT. Mechanisms of MEOX1 and MEOX2 regulation of the cyclin dependent kinase inhibitors p21 and p16 in vascular endothelial cells. PloS one. 2011;6:e29099.doi 10.1371/journal.pone.0029099
117. Zhou Y, Xia M, Cui C, Wei H, Jiang S, Peng J. Circulating Exosomal miR-221 from Maternal Obesity Inhibits Angiogenesis via Targeting Angptl2. International journal of molecular sciences. 2021 22.doi:10.3390/ijms221910343
118. Golson ML, Kaestner KH. Epigenetics in formation, function, and failure of the endocrine pancreas. Molecular metabolism. 2017;6:1066-76.doi 10.1016/j.molmet.2017.05.015
119. Moullé VS, Parnet P. Effects of Nutrient Intake during Pregnancy and Lactation on the Endocrine Pancreas of the Offspring. Nutrients. 2019 11.doi:10.3390/nu11112708
120. Bonner-Weir S, Aguayo-Mazzucato C, Weir GC. Dynamic development of the pancreas from birth to adulthood. Upsala journal of medical sciences. 2016;121:155-8.doi 10.3109/03009734.2016.1154906
121. Gregorio BM, Souza-Mello V, Mandarim-de-Lacerda CA, Aguila MB. Maternal high-fat diet is associated with altered pancreatic remodelling in mice offspring. European journal of nutrition. 2013;52:759-69.doi 10.1007/s00394-012-0382-9
122. Hill DJ, Hill TG. Maternal diet during pregnancy and adaptive changes in the maternal and fetal pancreas have implications for future metabolic health. Frontiers in endocrinology. 2024;15:1456629.doi 10.3389/fendo.2024.1456629
123. Saline M, Badertscher L, Wolter M, Lau R, Gunnarsson A, Jacso T. et al. AMPK and AKT protein kinases hierarchically phosphorylate the N-terminus of the FOXO1 transcription factor, modulating interactions with 14-3-3 proteins. The Journal of biological chemistry. 2019;294:13106-16.doi 10.1074/jbc.RA119.008649
124. Sebo ZL, Jeffery E, Holtrup B, Rodeheffer MS. A mesodermal fate map for adipose tissue. Development (Cambridge, England). 2018 145.doi:10.1242/dev.166801
125. Lecoutre S, Petrus P, Rydén M, Breton C. Transgenerational Epigenetic Mechanisms in Adipose Tissue Development. Trends in endocrinology and metabolism: TEM. 2018;29:675-85.doi 10.1016/j.tem.2018.07.004
126. Zhu Z, Cao F, Li X. Epigenetic Programming and Fetal Metabolic Programming. Frontiers in endocrinology. 2019;10:764.doi 10.3389/fendo.2019.00764
127. Kunte P, Barberio M, Tiwari P, Sukla K, Harmon B, Epstein S. et al. Neonatal adiposity is associated with microRNAs in adipocyte-derived extracellular vesicles in maternal and cord blood, a discovery analysis. International Journal of Obesity. 2024;48:403-13.doi 10.1038/s41366-023-01432-z
128. Ferland-McCollough D, Fernandez-Twinn DS, Cannell IG, David H, Warner M, Vaag AA. et al. Programming of adipose tissue miR-483-3p and GDF-3 expression by maternal diet in type 2 diabetes. Cell death and differentiation. 2012;19:1003-12.doi 10.1038/cdd.2011.183
129. Zhang J, Huang Y, Shao H, Bi Q, Chen J, Ye Z. Grape seed procyanidin B2 inhibits adipogenesis of 3T3-L1 cells by targeting peroxisome proliferator-activated receptor γ with miR-483-5p involved mechanism. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2017;86:292-6.doi 10.1016/j.biopha.2016.12.019
130. Li G, Fu S, Chen Y, Jin W, Zhai B, Li Y. et al. MicroRNA-15a Regulates the Differentiation of Intramuscular Preadipocytes by Targeting ACAA1, ACOX1 and SCP2 in Chickens. International journal of molecular sciences. 2019 20.doi:10.3390/ijms20164063
131. Zou Z, Liu X, Yu J, Ban T, Zhang Z, Wang P. et al. se in mice.Nuclear miR-204-3p mitigates metabolic dysfunction-associated steatotic liver disea. Journal of hepatology. 2024;80:834-45.doi 10.1016/j.jhep.2024.01.029
132. Shah KB, Chernausek SD, Teague AM, Bard DE, Tryggestad JB. Maternal diabetes alters microRNA expression in fetal exosomes, human umbilical vein endothelial cells and placenta. Pediatric research. 2021;89:1157-63.doi 10.1038/s41390-020-1060-x
133. Jiang S, Teague AM, Tryggestad JB, Aston CE, Lyons T, Chernausek SD. Effects of maternal diabetes and fetal sex on human placenta mitochondrial biogenesis. Placenta. 2017;57:26-32.doi 10.1016/j.placenta.2017.06.001
134. Shah KB, Chernausek SD, Garman LD, Pezant NP, Plows JF, Kharoud HK. et al. Human Milk Exosomal MicroRNA: Associations with Maternal Overweight/Obesity and Infant Body Composition at 1 Month of Life. Nutrients. 2021 13.doi:10.3390/nu13041091
135. Nelson BN, Friedman JE. Developmental Programming of the Fetal Immune System by Maternal Western-Style Diet: Mechanisms and Implications for Disease Pathways in the Offspring. International journal of molecular sciences. 2024 25.doi:10.3390/ijms25115951
136. Jefcoate CR, Larsen MC, Song YS, Maguire M, Sheibani N. Defined Diets Link Iron and α-Linolenic Acid to Cyp1b1 Regulation of Neonatal Liver Development Through Srebp Forms and LncRNA H19. International journal of molecular sciences. 2025 26.doi:10.3390/ijms26052011
137. Sugino KY, Mandala A, Janssen RC, Gurung S, Trammell M, Day MW. et al. Western diet-induced shifts in the maternal microbiome are associated with altered microRNA expression in baboon placenta and fetal liver. Frontiers in clinical diabetes and healthcare. 2022; 3.doi:10.3389/fcdhc. 2022 945768
138. Puppala S, Li C, Glenn JP, Saxena R, Gawrieh S, Quinn A. et al. Primate fetal hepatic responses to maternal obesity: epigenetic signalling pathways and lipid accumulation. The Journal of physiology. 2018;596:5823-37.doi 10.1113/jp275422
139. Zhang L, Zou W, Zhang S, Wu H, Gao Y, Zhang J. et al. Maternal high-fat diet orchestrates offspring hepatic cholesterol metabolism via MEF2A hypermethylation-mediated CYP7A1 suppression. Cellular & molecular biology letters. 2024;29:154.doi 10.1186/s11658-024-00673-8
140. Banfi C, Baetta R, Barbieri SS, Brioschi M, Guarino A, Ghilardi S. et al. Prenylcysteine oxidase 1, an emerging player in atherosclerosis. Communications biology. 2021;4:1109.doi 10.1038/s42003-021-02630-z
141. Kuan YC, Hashidume T, Shibata T, Uchida K, Shimizu M, Inoue J. et al. Heat Shock Protein 90 Modulates Lipid Homeostasis by Regulating the Stability and Function of Sterol Regulatory Element-binding Protein (SREBP) and SREBP Cleavage-activating Protein. The Journal of biological chemistry. 2017;292:3016-28.doi 10.1074/jbc.M116.767277
142. Simino LAP, Panzarin C, Fontana MF, de Fante T, Geraldo MV, Ignácio-Souza LM. et al. MicroRNA Let-7 targets AMPK and impairs hepatic lipid metabolism in offspring of maternal obese pregnancies. Scientific reports. 2021;11:8980.doi 10.1038/s41598-021-88518-8
143. Razo-Azamar M, Nambo-Venegas R, Quevedo IR, Juárez-Luna G, Salomon C, Guevara-Cruz M. et al. Early-Pregnancy Serum Maternal and Placenta-Derived Exosomes miRNAs Vary Based on Pancreatic β-Cell Function in GDM. The Journal of clinical endocrinology and metabolism. 2024;109:1526-39.doi 10.1210/clinem/dgad751
144. Luo X, Huang B, Xu P, Wang H, Zhang B, Lin L. et al. The Placenta Regulates Intrauterine Fetal Growth via Exosomal PPARγ. Advanced science (Weinheim, Baden-Wurttemberg, Germany). 2025: e2404983.doi:10.1002/advs.202404983.
145. Zhao H, Shang Q, Pan Z, Bai Y, Li Z, Zhang H. et al. Exosomes From Adipose-Derived Stem Cells Attenuate Adipose Inflammation and Obesity Through Polarizing M2 Macrophages and Beiging in White Adipose Tissue. Diabetes. 2018;67:235-47.doi 10.2337/db17-0356
146. Li Q, Hu W, Huang Q, Yang J, Li B, Ma K. et al. MiR146a-loaded engineered exosomes released from silk fibroin patch promote diabetic wound healing by targeting IRAK1. Signal transduction and targeted therapy. 2023;8:62.doi 10.1038/s41392-022-01263-w
147. Wang B, Pi Q, Mohsin A, Gao WQ, Guo M, Xu H. Exosomal miR-1246 of adipose stem cells attenuates obesity by polarizing M2 macrophages, reducing fat mass, and beiging of white adipose tissue. J Adv Res. 2025 doi:10.1016/j.jare.2025.05.049
148. Bi Y, Qiao X, Cai Z, Zhao H, Ye R, Liu Q. et al. Exosomal miR-302b rejuvenates aging mice by reversing the proliferative arrest of senescent cells. Cell metabolism. 2025;37:527-41.e6.doi 10.1016/j.cmet.2024.11.013
149. Luo X, Huang B, Xu P, Wang H, Zhang B, Lin L. et al. The Placenta Regulates Intrauterine Fetal Growth via Exosomal PPARγ. Advanced science (Weinheim, Baden-Wurttemberg, Germany). 2025;12:e2404983.doi 10.1002/advs.202404983
150. Zhang B, Tan L, Yu Y, Wang B, Chen Z, Han J. et al. Placenta-specific drug delivery by trophoblast-targeted nanoparticles in mice. Theranostics. 2018;8:2765-81.doi 10.7150/thno.22904
151. Beards F, Jones LE, Charnock J, Forbes K, Harris LK. Placental Homing Peptide-microRNA Inhibitor Conjugates for Targeted Enhancement of Intrinsic Placental Growth Signaling. Theranostics. 2017;7:2940-55.doi 10.7150/thno.18845
152. Della Pelle G, Markelc B, Čerček U, Živič U, Coupard M, Šribar J. et al. Novel vector for efficient siRNA delivery to lymphoblasts and melanoma based on genipin-spermine nanocarriers protected with hybrid erythrocyte membrane coating. Biomaterials advances. 2026;180:214537.doi 10.1016/j.bioadv.2025.214537
153. Yang J, Yun X, Zheng W, Zhang H, Yan Z, Chen Y. et al. Nanoscale engineered exosomes for dual delivery of Sirtuin3 and insulin to ignite mitochondrial recovery in myocardial ischemia-reperfusion. Journal of nanobiotechnology. 2025;23:439.doi 10.1186/s12951-025-03474-z
154. Rodríguez-Morales B, Antunes-Ricardo M, González-Valdez J. Exosome-Mediated Insulin Delivery for the Potential Treatment of Diabetes Mellitus. Pharmaceutics. 2021 13.doi:10.3390/pharmaceutics13111870
155. Zamanian MH, Norooznezhad AH, Hosseinkhani Z, Hassaninia D, Mansouri F, Vaziri S. et al. Human placental mesenchymal stromal cell-derived small extracellular vesicles as a treatment for severe COVID-19: A double-blind randomized controlled clinical trial. Journal of extracellular vesicles. 2024;13:e12492.doi 10.1002/jev2.12492
156. Kalluri VS, Smaglo BG, Mahadevan KK, Kirtley ML, McAndrews KM, Mendt M. et al. Engineered exosomes with Kras(G12D) specific siRNA in pancreatic cancer: a phase I study with immunological correlates. Nature communications. 2025;16:8696.doi 10.1038/s41467-025-63718-2
157. Rao J, Chen Y, Huang J, Wu R, Dong Z, Gao Y. et al. Inhibiting miR-182-3p Alleviates Gestational Diabetes Mellitus by Improving Insulin Resistance in Skeletal Muscle. Balkan medical journal. 2022;39:121-9.doi 10.4274/balkanmedj.galenos.2021.2021-8-140
158. Liu Y, Zhong C, Chen S, Xue Y, Wei Z, Dong L. et al. Circulating exosomal mir-16-2-3p is associated with coronary microvascular dysfunction in diabetes through regulating the fatty acid degradation of endothelial cells. Cardiovascular diabetology. 2024;23:60.doi 10.1186/s12933-024-02142-0
159. Chen X, Tao X, Wang M, Cannon RD, Chen B, Yu X. et al. Circulating extracellular vesicle-derived miR-1299 disrupts hepatic glucose homeostasis by targeting the STAT3/FAM3A axis in gestational diabetes mellitus. Journal of nanobiotechnology. 2024;22:509.doi 10.1186/s12951-024-02766-0
160. Wang J, Yao Y, Wang K, Li J, Chu T, Shen H. MicroRNA-148a-3p alleviates high glucose-induced diabetic retinopathy by targeting TGFB2 and FGF2. Acta diabetologica. 2020;57:1435-43.doi 10.1007/s00592-020-01569-7
161. Liu XL, Cao HX, Wang BC, Xin FZ, Zhang RN, Zhou D. et al. miR-192-5p regulates lipid synthesis in non-alcoholic fatty liver disease through SCD-1. World journal of gastroenterology. 2017;23:8140-51.doi 10.3748/wjg.v23.i46.8140
162. Wang G, Jing SY, Liu G, Guo XJ, Zhao W, Jia XL. et al. miR-99a-5p: A Potential New Therapy for Atherosclerosis by Targeting mTOR and Then Inhibiting NLRP3 Inflammasome Activation and Promoting Macrophage Autophagy. Disease markers. 2022;2022:7172583.doi 10.1155/2022/7172583
163. Li R, Tao H, Pan K, Li R, Guo Z, Chen X. et al. Extracellular vesicles derived from mesenchymal stem cells alleviate renal fibrosis via the miR-99b-5p/mTOR/autophagy axis in diabetic kidney disease. Stem cell research & therapy. 2025;16:142.doi 10.1186/s13287-025-04265-x
164. Liu H, Wen J, Tian X, Li T, Zhao J, Cheng J. et al. miR-125a-3p regulates the expression of FSTL1, a pro-inflammatory factor, during adipogenic differentiation, and inhibits adipogenesis in mice. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2023;37:e23146.doi 10.1096/fj.202300851R
165. Zhang Y, Li W, Li H, Zhou M, Zhang J, Fu Y. et al. Circ_USP36 Silencing Attenuates Oxidized Low-Density Lipoprotein-Induced Dysfunction in Endothelial Cells in Atherosclerosis Through Mediating miR-197-3p/ROBO1 Axis. Journal of cardiovascular pharmacology. 2021;78:e761-e72.doi 10.1097/fjc.0000000000001124
166. Li W, Yuan X, He X, Yang L, Wu Y, Deng X. et al. The downregulation of miR-22 and miR-372 may contribute to gestational diabetes mellitus through regulating glucose metabolism via the PI3K/AKT/GLUT4 pathway. Journal of clinical laboratory analysis. 2022;36:e24557.doi 10.1002/jcla.24557
167. Xue L, Xiong C, Li J, Ren Y, Zhang L, Jiao K. et al. miR-200-3p suppresses cell proliferation and reduces apoptosis in diabetic retinopathy via blocking the TGF-β2/Smad pathway. Bioscience reports. 2020 40.doi:10.1042/bsr20201545
168. Zheng Z, Mo J, Lin F, Wang J, Chen J, Luo H. et al. Milk Exosomes from Gestational Diabetes Mellitus (GDM) and Healthy Parturient Exhibit Differential miRNAs Profiles and Distinct Regulatory Bioactivity on Hepatocyte Proliferation. Molecular nutrition & food research. 2023;67:e2300005.doi 10.1002/mnfr.202300005
169. Majumdar S, Chowdhury DR, Chakraborty BC, Chowdhury A, Datta S, Banerjee S. MiR-451a attenuates hepatic steatosis and hepatitis C virus replication by targeting glycerol kinase. Journal of translational medicine. 2025;23:322.doi 10.1186/s12967-025-06286-9
170. Zhang Q, Ye X, Xu X, Yan J. Placenta-derived exosomal miR-135a-5p promotes gestational diabetes mellitus pathogenesis by activating PI3K/AKT signalling pathway via SIRT1. Journal of cellular and molecular medicine. 2023;27:3729-43.doi 10.1111/jcmm.17941
Author contact
Corresponding authors: Jieying Liu (Email: liujieying163.com) and Xinhua Xiao (Email: xiaoxh2014163.com).

 Global reach, higher impact
Global reach, higher impact