Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2017; 13(2):157-166. doi:10.7150/ijbs.17440 This issue Cite
Research Paper
Regulatory Axis of miR-195/497 and HMGA1-Id3 Governs Muscle Cell Proliferation and Differentiation
1. Shenzhen Key Laboratory of Microbial Genetic Engineering, College of Life Sciences, Shenzhen University, Shenzhen, Guangdong, 518060, China.
2. Key Laboratory of Optoelectronic Devices and Systems of Ministry of Education and Guangdong Province, College of Optoelectronic Engineering, Shenzhen University, Shenzhen, Guangdong, 518060, China.
3. Department of Physiology, Shenzhen University Health Science Center, Shenzhen, Guangdong, 518000, China.
* These authors contributed equally to this work.
Abstract
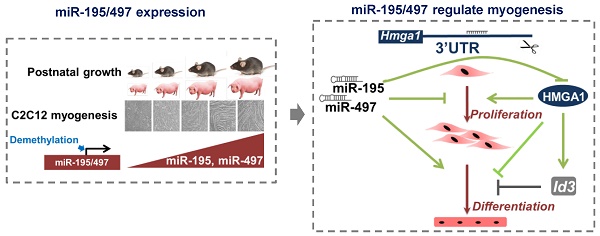
Myocytes withdraw from the cell cycle to differentiate during muscle development. Given the capacity of microRNAs (miRNAs) to regulate gene expression during development, we screened for miRNAs that were associated with muscle development. S-Poly(T) Plus analysis of 273 miRNAs in porcine longissimus dorsi muscles revealed 14 miRNAs that were strongly upregulated with age of postnatal muscle development in vivo, including miR-195 and miR-497. These two miRNAs were also strongly upregulated at late differentiation stages of mouse skeletal myoblast C2C12 cells, and demethylation treatment induced significant upregulation of miR-195/497. Manipulation of miR-195/497 expression resulted in dramatic changes in the proliferation and differentiation of C2C12 cells. We identified high-mobility group AT-hook 1 (Hmga1) mRNA as a highly conserved target of miR-195/497 in C2C12 myoblasts. Overexpression of miR-195/497 or Hmga1 silencing in C2C12 cells promoted myogenic differentiation. Moreover, we showed that miR-195/497 repressed Hmga1, which in turn downregulated one of the HMGA1 downstream targets Id3, whose inhibitory effect on myogenic differentiation is well established. Our study revealed a subset of potential development-associated miRNAs and suggests a novel regulatory axis for myogenesis in which miR-195/497 promote myogenic differentiation by repressing the HMGA1-Id3 pathway.
Keywords: miR-195/497, Hmga1, myoblast, skeletal muscle, differentiation, proliferation.

 Global reach, higher impact
Global reach, higher impact