Impact Factor ISSN: 1449-2288
Int J Biol Sci 2018; 14(2):147-155. doi:10.7150/ijbs.23231 This issue Cite
Research Paper
NLRP12 Promotes Mouse Neutrophil Differentiation through Regulation of Non-canonical NF-κB and MAPKERK1/2 Signaling
1. Department of Oncology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China;
2. Department of Chemotherapy, Nanjing Medical University Affiliated Cancer Hospital, Cancer Institute of Jiangsu Province, Nanjing, Jiangsu, China;
3. Department of Radiation Oncology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China.
4. Department of Cell and Developmental Biology, University of Illinois at Urbana-Champaign, Urbana, IL, USA.
5. Department of Burns and Plastic Surgery, Shanghai Ninth People's Hospital, Shanghai JiaoTong University School of Medicine, Shanghai, China.
*These authors have contributed equally to this work
Received 2017-10-9; Accepted 2017-12-9; Published 2018-1-13
Abstract
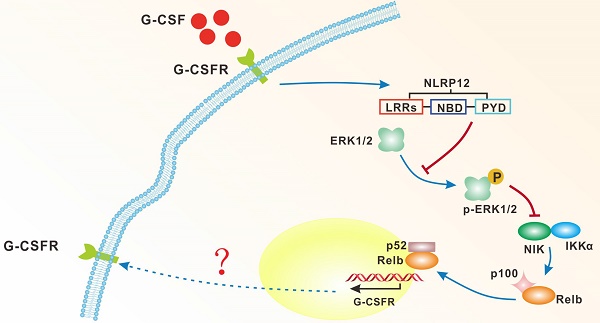
Neutrophils are the most important component of the innate immune system. Mechanistic understanding of the mechanism underlying neutrophil differentiation remains elusive. Using genome-wide RNA-seq, we identified genes whose expression is dramatically up-regulated during neutrophil differentiation. Among them is nucleotide-binding leucine-rich repeat and pyrindomain-containing receptor 12 (NLRP12), which plays a role in immune inflammatory responses. Genetic ablation of NLRP12 suppresses NF-κB inducing kinase (NIK) stabilization, RelB nuclear translocation and neutrophil differentiation in vitro. At a mechanistic level, NLRP12 inhibits the activity of mitogen-activated protein kinases (MAPK)/extracellular signal-regulated kinases (ERK1/2), relieves ERK1/2 suppression of NIK protein levels. Thus, NLRP12 enhances noncanonical NF-κB signaling through inhibition of ERK1/2 signaling, thereby promoting neutrophil differentiation.
Keywords: myeloid progenitors, NLRP12, neutrophils differentiation, NF-κB, ERK1/2
Introduction
Neutrophils are the most abundant type of granulocytes and the most abundant type of white blood cells. These cells are multifunctional, short-lived and highly mobile[1, 2]. As the most important part of the innate immune system, neutrophils are indispensable for the destruction of microorganisms[1, 3-5]. Neutrophils are produced from hematopoietic stem cells (HSCs) through myeloid differentiation, and abnormal neutrophil differentiation can lead to the occurrence of various blood diseases, such as leukemia, myeloid aplasia, and dysplasia[6-8]. Therefore, a better understanding of the mechanism underlying neutrophil differentiation has long-standing significances.
Many in vitro models have been used to study neutrophil differentiation, including primary normal bone marrow precursors and leukemic cells[9, 10]. Recently, embryonic stem cells (ESCs) with the ability to differentiate into nearly all cell types, have been used to produce functional neutrophil cells[11]. From ESCs we generated a new myeloid progenitor cell line (named as mEB8-ER) immortalized through ectopic expression of β-estradiol-regulated-Hoxb8 protein (Hoxb8-ER). The mEB8-ER exhibits normal karyotyping, thus overcoming the limitation of the commonly used neutrophil differentiation models and can differentiate into functional neutrophils in several days. Using the model, we previously identified that mammalian target of rapamycin complex 1 (mTORC1) as an important regulator of neutrophil differentiation[11].
In this study, we sought to further dissect the mechanism for neutrophil differentiation. We exploited genome-wide RNA-seq analysis to identify genes differentially expressed during neutrophil differentiation. With this screen, we identified NLRP12 as a critical regulator of neutrophil differentiation. NLRP12 is up-regulated during neutrophil differentiation, while depletion of NLRP12 inhibits neutrophils differentiation. NLRP12 promotes neutrophil differentiation by enhancing noncanonical NF-κB signaling through inhibition of ERK1/2 activities.
Results
NLRP12 regulates neutrophil differentiation of myeloid progenitors
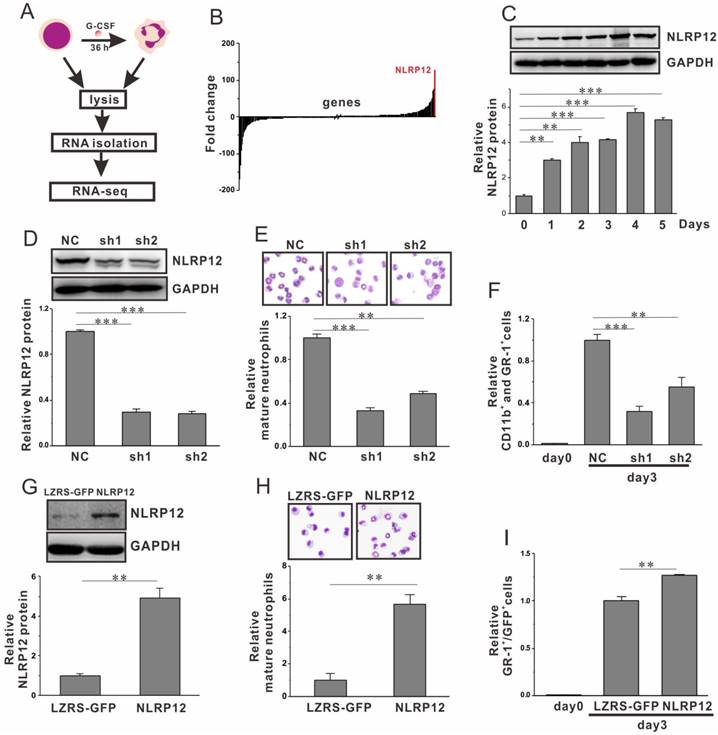
The myeloid progenitors (mEB8-ER) derived from mouse embryonic stem cell (mESCs) can differentiate into neutrophils in the presence of cytokine G-CSF, which has been proved as a good research model for dissecting neutrophil differentiation[11]. Using this model, we exploited RNA-Seq to identify genes involved in the differentiation process (Fig. 1A). We calculated the fold of enrichment or reduction of each gene`s RNA level between the myeloid progenitors cells and those undergoing neutrophil differentiation with G-CSF treatment for 36 hours (Fig. 1B). As expected, most of the genes exhibited little difference between myeloid progenitors and differentiating neutrophils. However, 103 genes were up-regulated more than 10-fold, and 69 genes were decreased more than 10-fold. We then chose several dramatically changed genes including Slc40a1, Slfn4, XDH, G0S2, Smarcad1, VPS33b and NLRP12 for subsequent studies. The expression levels of the genes during differentiation were traced by q-PCR and western blot. Expectedly, upon G-CSF stimulation, the levels of the genes changed over time in line with the result from RNA-Seq (Fig. 1C, Fig. S1). Under the condition of RNA interference or gene ectopic expression, we induced mEB8-ER cells to undergo differentiation in the presence of G-CSF and determined the expression of differentiation markers, such as GR-1 and CD11b.We found that most genes exerted little effect on the differentiation process, except NLRP12 (Figs. S2A-C or data not shown). The role of NLRP12 was further explored (the rest of the study), whereas analysis of other genes awaits future experimentation.
Regulation of the levels of NLRP12 (the most enriched one, 129-fold, Fig. 1B) can cause an obvious effect on the differentiation process as evident by detecting pan-myeloid marker CD11b and murine neutrophil marker GR-1 using q-PCR (Figs. S2B-C). For the studies, two shRNAs targeting NLRP12 (sh1 and sh2) were introduced to mEB8-ER cells by retrovirus-mediated infection. The expression of NLRP12 was successfully depleted as revealed by western blotting and q-PCR (Fig. 1D, Fig. S2A). After 3 days of induction with G-CSF, we examined the morphology and the nucleo-cytoplasmic ratio by using Wright-Giemsa staining. In the control group, approximately 65% of the mEB8-ER progenitors differentiated directionally into neutrophils (Fig. 1E). In contrast, with NLRP12 depletion the ratio of differentiated neutrophils were significantly reduced (21% and 31% in sh1 and sh2 treated groups, respectively). We further confirmed these results by detecting CD11b and GR-1 using flow cytometry (Fig. 1F, Fig. S1D). The mEB8-ER cells were detected with a relatively lower percent of GR-1 and CD11b positive cells with NLRP12 depletion than the control cells. In keeping with the results from NLRP12 depletion, ectopic expression of NLRP12 (Fig. 1G, Fig. S2E) enhanced neutrophil differentiation activities, as revealed by Wright-Giemsa staining and flow cytometry (Fig. 1H and I). Thus, NLRP12 serves as a critical regulator of neutrophil differentiation.
NLRP12 enhances noncanonical NF-κB signaling
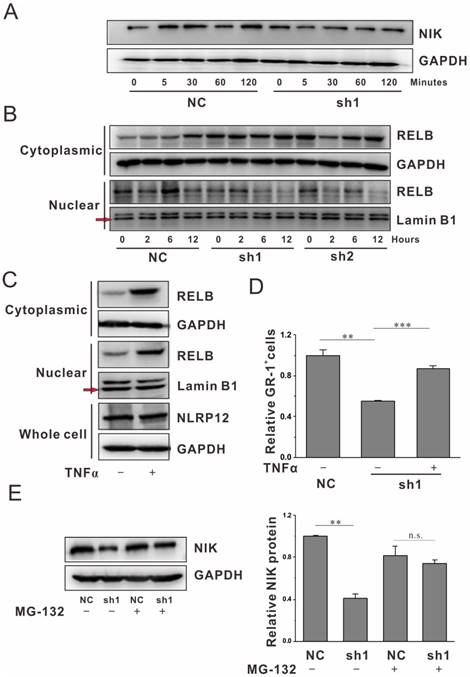
Previous in vitro studies have shown that NLRP12 can interact with NF-κB inducing kinase (NIK) leading to its degradation. NIK plays multiple roles in the regulation of noncanonical NF-κB signaling in monocytes, and osteoclast formation[12-14]. Thus, we tested whether inhibition of NLRP12 affects the noncanonical NF-κB pathway in neutrophils during differentiation. As shown in Fig. 2A, NLRP12 depletion attenuates the expression level of NIK at each time points examined during the differentiation. RelB is a key noncanonical NF-κB pathway subunit downstream of NIK[15]. Inhibition of NLRP12 significantly decreased translocation of RelB into the nuclear compartment, indicating that noncanonical NF-κB signaling is suppressed when NLRP12 is depleted (Fig. 2B).
We next performed a rescue experiment by activating the noncanonical NF-κB pathway with TNFα. TNFα is a pro-inflammatory cytokines that can initiate the intracellular signal transduction pathways, ultimately leading to activation of downstream transcription factors, including NF-κB[16]. TNFα remarkably increased the nuclear translocation of Relb (Fig. 2C), suggesting the activation of noncanonical NF-κB pathway by TNFα, but did not alter the level of NLRP12. The ratio of GR-1+cells was significantly increased in NLRP12-depleted cells in the presence of TNFα (Fig. 2D). These results suggest that the NF-κB pathway serves as a bona fide target of NLRP12 to control neutrophil differentiation induced by G-CSF.
To dissect how NLRP12 might regulate NIK expression, we first examined the mRNA level of NIK with NLRP12 depletion during neutrophil differentiation, but detected no significant changes (Fig. S3A). To ask whether NLRP12 regulates NIK at the protein level, we carried out a proteasome inhibition assay by applying MG-132, a well-known inhibitor of proteasome that displays great potency in inhibiting the degradation of proteasomes[17]. The addition of MG-132 drastically increased the protein level of NIK in NLRP12-depleted cells (Fig. 2E).
Taken together, these results suggest that NLRP12 enhances neutrophil differentiation and does so by enhancing the noncanonical NF-κB pathway through protection of NIK from proteasomal degradation.
NLRP12 regulates neutrophil differentiation from myeloid progenitors. (A) Schematic outline of a RNA-seq experiment to determine the genes that involved in neutrophil differentiation. (B) Increased or decreased genes that were screened as described in (A). (C) The expression levels of NLRP12 during neutrophil differentiation were analyzed by western blotting. (D-F) Two shRNAs target NLRP12 were introduced into the mEB8-ER cells, the cells were induced to differentiate in the presence of G-CSF (2 ng/mL, 3 days). The expression of NLRP12 in mEB8-ER cells with or without NLRP12 depletion was analyzed by western blotting (D). The differentiation level was test by Wright-Giemsa staining of the differentiated cells (E) and flow cytometry for CD11b+ and GR-1+ (F). (G) The expression of NLRP12 in mEB8 cells with or without NLRP12 overexpression was analyzed by western blotting. (H-I) The cells with or without NLRP12 overexpression were induced to differentiate in the presence of G-CSF (2ng/mL, 3 days). The differentiation level was test by Wright-Giemsa staining of the differentiated cells (H) and flow cytometry for GR-1+/GFP+ cells (I). Three separate experiments were conducted, and quantification of 3 replicates of a typical experiment. Each bar represents the mean ± SEM (error bars). All values were normalized to the level (=1) in control group. **p< 0.01 and ***p < 0.001.
NLRP12 inhibits ERK1/2 signaling during neutrophil differentiation
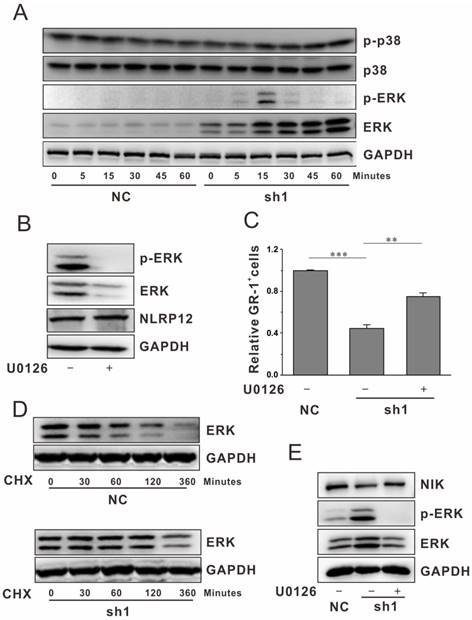
The MAPK pathway plays an important role in numerous cellular events, including cell differentiation[18]. Moreover, some Nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs) have been shown to function as activators of MAPK transcription factors[19, 20]. Therefore, we also investigated whether NLRP12 affects the MAPK pathway during neutrophil differentiation. The phosphorylation and total protein levels of ERK1/2, JNK and p38 were examined by western blotting. The levels of total ERK1/2 and ERK1/2 phosphorylation were significant elevated in NLRP12-depleted cells compared to the control cells (Fig. 3A). But there were little changes of total p38 and p38 phosphorylation with NLRP12-depletion. However, we did not detect the signal of JNK protein and phospho-JNK in these cells. The results suggest that the ERK1/2 pathway might act as downstream targets of NLRP12 during neutrophil differentiation.
NLRP12 enhances noncanonical NF-κB signaling. (A-B) mEB8-ER cells with or without NLRP12 depletion were stimulated with G-CSF (2 ng/mL) for the times indicated. The expression of NIK in the whole cell lysates was analyzed by western blotting (A). Extracts from the nuclear and cytoplasmic fractions were analyzed by western blotting for RelB (B). (C) mEB8-ER cells treated with or without TNFα (10 ng/mL) were induced to differentiate in the presence of G-CSF, and extracts from the nuclear and cytoplasmic fractions were analyzed by western blotting for RelB. (D) Relative percentage of GR-1+ cells in the mEB8-ER cells with or without TNFα treatment, NLRP12 depletion, was measured by flow cytometry. (E) The expression of NIK in the mEB8 cells with or without MG-132 treatment, NLRP12 depletion, was measured by western blotting. Three separate experiments were conducted, and quantification of 3 replicates of a typical experiment. Each bar represents the mean ±SEM (error bars). All values were normalized to the level (=1) in cells of control group. **p< 0.01 and ***p < 0.001.
To ask whether the ERK1/2 pathways function in neutrophil differentiation, we examined the effect of ERK1/2 inhibition. U0126 is a highly selective inhibitor of p-ERK1/2. Treatment of cells with U0126 markedly reduced the total and the activated levels of ERK1/2, suggesting inhibition of ERK1/2 pathway. Moreover, no significantly change of NLRP12 level was observed with the inhibitor treatments (Fig. 3B).
To explore the function of ERK1/2 in NLRP12-mediated neutrophil differentiation, we performed rescue experiments by inhibiting ERK1/2 pathway in cells with NLRP12 depletion. In NLRP12 depleted cells, the ratio of GR-1+ cells was significantly increased in the presence of U0126 (Fig. 3C). Taken together, these results suggest that ERK1/2 acts as a downstream functional target of NLRP12.
It was worth noting that NLRP12 inhibition also increased the total level of ERK1/2. We did not detect significant changes in the mRNA level of ERK1/2 in cells with NLRP12 depletion during neutrophil differentiation (Fig. S3B). We then carried out a protein degradation assay using cycloheximide (CHX) to determine potential proteolysis of ERK1/2 during differentiation. The proteolysis of ERK1/2 was significantly suppressed after NLRP12 depletion, as evidenced by the increase of the half-life of proteolysis from 1 h to more than 6 h after treatment (Fig. 3D, Fig. S3C). Thus, NLRP12 inhibits the ERK1/2 pathway, most likely by promoting the proteolysis of ERK1/2 protein instead of down-regulating the ERK1/2 transcripts.
Because NLRP12 plays a critical role in neutrophil differentiation by either inhibiting the ERK1/2 activity or enhancing the noncanonical NF-κB pathway, we next determined the relationship between ERK1/2, and NF-κB signaling during this process. As shown in Fig. 3E, U0126 markedly increased the protein level of NIK in NLRP12-depleted mEB8-ER cells, thus indicating that the ERK1/2 pathway negatively regulates NIK expression during neutrophil differentiation.
G-CSFR is down-regulated in NLRP12-depleted neutrophils
Multiple regulatory factors and diverse signal transduction pathways have been implicated in neutrophil differentiation[21]. In particular, C/EBPα is considered a master regulator of neutrophil differentiation[22]. Extensive studies have also implicated the transcription factors PU.1 in proper differentiation of neutrophils[23]. G-CSF acts by binding to its receptor (G-CSFR), a member of the class I cytokine receptor superfamily expressed on myeloid progenitors, causing phosphorylation of intracellular tyrosines and activation of signal transduction pathways[24]. In this study, we detected the expression of G-CSFR, PU.1, C/EBPα. As shown in Figs. S4A and B, depletion of NLRP12 did not cause obvious changes in the level of PU.1 and CEBPα, but led to significant down-regulation of G-CSFR. The addition of TNFα to cells with NLRP12 depletion markedly increased the mRNA level of G-CSFR (Fig. S4C). The similar result was also observed with U1026 treatment (Fig. S4D). Thus, NLRP12 activates the NF-κB pathway, which may in turn enhances the level of G-CSFR and consequently G-CSFR-mediated myeloid differentiation. Further works are needed to be done to detail the relationship between G-CSFR and NLRP12.
NLRP12 inhibits MAPK activation. (A) mEB8-ER cells with or without NLRP12 depletion were stimulated with G-CSF (2 ng/mL) for the times indicated. The phosphorylation and total expression levels of ERK1/2 and p38 in the whole cell lysates were analyzed by western blotting. (B) mEB8-ER cells were treated with U0126 (10 μM), and the expressions levels of NLRP12, p-ERK1/2 and ERK1/2, were analyzed by western blotting. (C) Relative percentage of GR-1+ cells in mEB8 cells with or without NLRP12 depletion and U0126 treatment were measured by flow cytometry. (D) The expression of ERK1/2, in the normal differentiated mEB8-ER cells and NLRP12 deficiency cells with CHX (1 μg/mL) treatment were analyzed by western blotting. (E) mEB8-ER cells with or without NLRP12 depletion were treated with 10 μM U0126. The cells were then induced to differentiate in the presence of G-CSF (2 ng/mL). The expression of NIK, p-ERK1/2 and ERK1/2 were analyzed using western blotting. Three separate experiments were conducted, and quantification of 3 replicates of a typical experiment. Each bar represents the mean ±SEM (error bars). All values were normalized to the level (=1) in cells of control group. **p< 0.01 and ***p < 0.001.
NLRP12 has a conserved function in primary HPCs
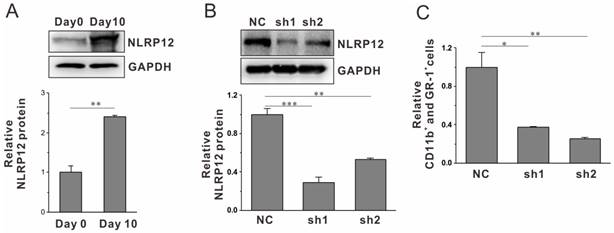
Our results from the mEB8-ER cell system suggest that NLRP12 is a critical regulator of neutrophil differentiation. To confirm the results in primary cells, we isolated Sca-1-negative and c-Kit-positive progenitors from the mouse bone marrow as previously reported[25]. The expression level of NLRP12 in the cells was dramatically increased during differentiation (Fig. 4A). The role of NLRP12 was further explored using RNAi (Fig. 4B). After 10 days of cultivation in G-CSF-containing medium, the number of the cells expressing CD11b and GR-1 was significantly reduced (Fig. 4C), confirming that NLRP12 is a critical regulator of neutrophil differentiation.
Discussion
In conclusion, by using our previously established myeloid progenitor cell model, we dissected the mechanism of neutrophil differentiation. Using genome-wide profiling, we find that NLRP12 is significantly up-regulated during neutrophil differentiation. RNAi-mediated depletion of NLRP12 suppressed neutrophils differentiation in both our cell model and primary HPCs. At a molecular level, NLRP12 enhances NIK stabilization and RelB nuclear translocation while inhibiting ERK1/2 signaling. We further show that ERK1/2 negatively regulates NIK during NLRP12-mediated neutrophil differentiation. Thus, NLRP12, when up-regulated rapidly after induction of neutrophil differentiation, promotes differentiation by inhibiting ERK1/2 signaling and consequently enhancing noncanonical NF-κB signaling.
Nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs) consist of a family of intracellular pattern-recognition receptors and have been widely investigated as signaling platforms that play important roles in regulation of inflammatory signaling in response to microbial signals and cellular stress[26-28]. NLRP12 is a recently identified member of NLRs predominately expressed in the myeloid/monocytic lineage, including macrophages, granulocytes, and immature dendritic cells. It has been proposed with various functions. NLRP12's function as a negative regulator of toll-like receptor (TLR)-induced cytokine production has been confirmed by in vitro studies[14, 29, 30]. NLRP12 was also found to regulate the neutrophil function, such as cell migration. NLRP12 worked as a negative regulator of directed neutrophil migration but not dendritic cells[31]. Moreover, NLRP12 has also shown to antagonize noncanonical NF-κB in monocytes[12]. Although a role of NLRP12 in the regulation of inflammatory responses in innate immune cells has been revealed, whether NLRP12 functions in production of immune cells is unclear. In the current study, by using genome-wide RNA-seq and follow-up functional analyses, we demonstrate, for the first time, that NLRP12 plays an essential role in neutrophil differentiation from myeloid progenitor cells.
NF-κB plays critical roles in regulating inflammatory and immune responses[32, 33]. In vitro and in vivo studies have demonstrated that NLRP12 serves as a negative regulator of NF-κB signaling[12, 34]. An interaction between NLRP12 and NIK stabilization in human macrophage cell lines has been revealed, and this interaction leads to proteasome-dependent degradation of NIK[12, 14]. As a result, activation of noncanonical NF-κB is suppressed. In contrast, the effect of NLRP12 on canonical NF-κB signaling has been reported to be modest by several studies[13, 14]. However, our studies suggest that NLRP12 functions as a positive regulator of NF-κB signaling during neutrophil differentiation. The noncanonical NF-κB signaling is suppressed when NLRP12 expression is reduced (Figs. 2A and B). Our data suggest that NLRP12 maintains the stability of NIK necessary for noncanonical NF-kB activities. The discrepancies between earlier studies and ours are possible due to the difference in cellular context, that is, NLRP12 may play an opposite role during granulopoiesis. Specifically, we find that NLRP12 inhibition promotes the proteasomal degradation of NIK. Interestingly, we also find that NLRP12 also affects canonical NF-κB signaling as evidenced by suppression of IκBα and p65 nuclear translocation upon NLRP12 inhibition (data not shown). It seems that the relationships of NLRP12 with NF-κB signaling during neutrophils differentiation are quite different from that in other cells.
Our studies also demonstrated that the ERK1/2 signaling serves as a negative regulator of neutrophil differentiation whose activity is inhibited by NLRP12. Our findings confirm and extend previous studies showing that sustained activation of the MAPKERK1/2 is essential for suppression of myeloid differentiation[35]. We also establish a functional link between NLRP12 and ERK1/2, demonstrating that the differentiation defect in NLRP12-depleted myeloid progenitors cells can be rescued by inhibition of the ERK1/2 pathway.
NLRP12 has a conserved function in primary HPCs.The expression levels of NLRP12 during neutrophil differentiation using Sca-1-negative and c-Kit-positive progenitors from the mouse bone marrow were analyzed by western blotting (A). Two shRNAs target NLRP12 were introduced into the cells during differentiation, and the expression of NLRP12 were analyzed by western blotting (B), relative percentage of CD11b+ and GR-1+ cells in each group were measured by flow cytometry (C). Three separate experiments were conducted, and quantification of 3 replicates of a typical experiment. Each bar represents the mean ±SEM (error bars). All values were normalized to the level (=1) in cells of control group. *p< 0.05, **p < 0.01 and ***p < 0.001.
The possible model of the mechanism of NLRP12-dependent neutrophils differentiation.
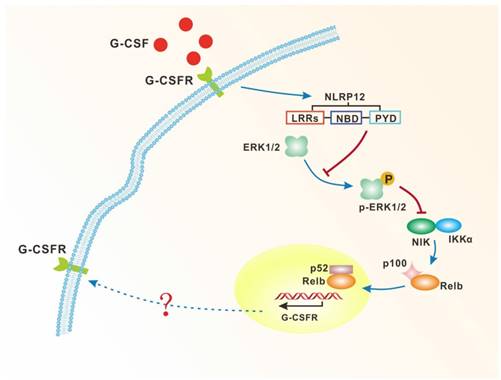
It has been documented that multiple members of the NLR family not only initiate proinflammatory NF-κB signaling, but also have a regulatory effect on MAPK signaling in response to extracellular stimuli[20]. A recent study demonstrated that NLRP12 inhibits NF-κB activation by dampening phosphorylation of IκBα and ERK1/2, resulting in the enhancement of intracellular bacterial survival[30]. However, an intriguing observation arising from our study is that NLRP12 positively regulates the NF-κB signaling while it has a negative regulatory effect on the ERK1/2 pathway. Moreover, pharmacological inhibition of ERK1/2 leads to enhanced accumulation of newly synthesized NIK (Fig. 3E).
In conclusion, our study has revealed a novel function of NLRP12 in the regulation of neutrophil differentiation. Our findings provide novel mechanistic insight into myeloid differentiation and may prove useful for controlling the neutrophil differentiation and possibility for the understanding of leukemia.
Material and methods
Cell culture and differentiation
mEB8-ER cells are sort of hematopoietic progenitor cells which were constructed in our previous study[11]. The cells were cultured in opti-mem medium (11058-021; GIBCO Inc.) containing penicillin sulphate (100 units/mL), streptomycin (100 μg/mL) (15140122; GIBCO Inc.), 10% fetal bovine serum (1527494; GIBCO Inc.), 20 ng/ml SCF (250-03; Peprotech), 1uM estradiol (E2758; Sigma-Aldrich) and 2 mM L-Glutamine (25030081; GIBCO Inc.) at 37°C and 5% CO2. For neutrophils differentiation, the mEB8 cells (~1×106) were incubated with 2ng/ml G-CSF (250-05; Peprotech) for 1.5, 3 or 5 days in the absence of SCF and estradiol. 293T cells were maintained in DMEM (SH30243.01B; Hyclone) supplemented with penicillin sulphate (100 units/mL), streptomycin (100 μg/mL), 10% FBS (1739463; GIBCO Inc.) and 2 mM L-Glutamine.
Isolation of murine bone marrow progenitors
Mouse bone marrow progenitor cells were isolated from femurs and tibias of C57BL/6 mice, cultured and expanded in medium containing 10 ng/mL IL-3 (213-13; Peprotech), 20 ng/mL IL-6 (216-16; Peprotech), and 25 ng/mL stem cell factor (SCF), as described previously[11]. The differentiation of the cells was performed by the adding of 20 ng/mL G-CSF for 10 days. The animal experiments were approved by the ethics committee of Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine.
shRNA-mediated gene silencing
shRNA-mediated gene silencing was carried out to construct NLRP12 knockdown cells using shRNA plasmids containing hairpin NLRP12 shRNA sequence. Viral packaging mix was used to cotransfect with the shRNA plasmids into 293T cells to produce NLRP12 shRNA viral particles. Gag-pol/VSVG retrovirus package was used for NLRP12 knockdown mEB8 cells and primary HCPs. The empty vector (pSIREN) was used as control. The cells were then infected with the viral particles followed by selection with 2 μg/mL puromycin (A610593; Sangon Biotech.), resulting in NLRP12 cell lines with different shRNA sequences inserted into the genome. Knockdown efficacy of the NLRP12 gene expression was determined by q-PCR and western blotting, and the highest knockdown efficacy were used in the follow-up experiments. The two shRNA sequences target NLRP12 are as follows: sh1: 5'-CACTCGGCTTCTCCTAGTAA-3'; sh2: 5'-GTAGATTCCACGCAGATAAT-3'.
q-PCR
Total RNA of mEB8 and primary HCPs were isolated using trizol (9018; Takara). First-strand cDNA was synthesized from 500ng of RNA using PrimeScript RT reagent Kit (RR037A; Takara). Real-time PCR assay for NLRP12, GR-1, CD11b, NIK, ERK1/2, and β-actin was performed using SYBR Green master mix (B21202; Bimake) on Roche PCR480 real-time PCR system. Simultaneous quantification of β-actin mRNA was used as an internal control. The sequences of q-PCR primers are listed in Table 1.
Sequences of q-PCR primers were used
| Primer | Sequence |
|---|---|
| β-actin-F | 5'-GGCTGTATTCCCCTCCATCG-3' |
| β-actin-R | 5'-CCAGTTGGTAACAATGCCATGT-3' |
| CD11b-F | 5'-CCATGACCTTCCAAGAGAATGC-3' |
| CD11b-R | 5'-ACCGGCTTGTGCTGTAGTC-3' |
| GR-1-F | 5'-GACTTCCTGCAACACAACTACC-3' |
| GR-1-R | 5'-ACAGCATTACCAGTGATCTCAGT-3' |
| NLRP12-F | 5'-GGATGGCCTCTATCGACTGTC-3' |
| NLRP12-R | 5'-CCTCTGCAATCCCCAGGAATAA-3' |
| GCSFR-F | 5'-CTGATCTTCTTGCTACTCCCCA-3' |
| GCSFR-R | 5'-GGTGTAGTTCAAGTGAGGCAG-3' |
| CEBPα-F | 5'-AAACAACGCAACGTGGAGA-3' |
| CEBPα-R | 5'-GCGGTCATTGTCACTGGTC-3' |
| PU.1-F | 5'-TCTTCTGCACGGGGAGACAG-3' |
| PU.1-R | 5'-GGACGAGAACTGGAAGGTACC-3' |
| NIK-F | 5'-AACGACTCTCCCGCGAAGA-3' |
| NIK-R | 5'-CGGCAGTGACCGTTTTAACA-3' |
| ERK-F | 5'-CAGGTGTTCGACGTAGGGC-3' |
| ERK-R | 5'-TCTGGTGCTCAAAAGGACTGA-3' |
Western blotting
The mEB8 and mouse bone marrow progenitor cells were lysed with RIPA buffer after twice washing with PBS. Then the proteins were quantified by BCA Protein Assay Kit. Primary antibodies used in western blotting were anti-NLRP12 (SAB3500555; Sigma-Aldrich), anti-NIK (AB191592; Abcam), anti-Relb (AB180127; Abcam), anti-ERK1/2 (9102; CST), anti-phospho-ERK1/2 (9101; CST), anti-p38 (9212; CST), anti-phospho-p38 (4511; CST), anti-JNK (9252; CST), anti-phospho-JNK (9255; CST), anti-Gapdh (HC301; Transgen Biotech.) and anti-laminb (66095; Proteintech). The blot was detected with horseradish-peroxidase-labeled anti-rabbit (7074S; CST) or anti-mouse secondary antibody (7076S; CST) and ECL Plus solution (WBKLS0500; Millipore).
Flow cytometry
Cells were incubated for 30 min at room temperature with 2 μg/mL of anti-mouse GR-1-FITC (108406; Biolegend), anti-mouse GR-1-PE (108408; Biolegend), anti-mouse CD11b-PE (101208; Biolegend) antibodies or isotypeIgG antibody (control). Cells were then washed and data were collected from ≥100,000 viable cells on a LSRII flow cytometer (Beckman) and post-acquisition analyses were performed with FlowJo software.
Giemsa staining
For morphologic analysis of the differentiated neutrophils, a well-established Giemsa staining assay was used (057K4355; Sigma-Aldrich). Images were collected using a microscopy (Nikon) and the mature neutrophils were counted using three independent images.
Statistical analysis
Results are shown as mean ± std. Statistical significance was determined using t-test. Results were considered significant when p<0.05.
Supplementary Material
Supplementary figures.
Acknowledgements
This work was supported by National Natural Science Foundation of China (grant81370600, D.L.); The Program for Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning (TP2015022, D.L.); Shanghai Pujiang Program (15PJ1404800, D.L.) and Innovation Program of Shanghai Municipal Education Commission (15ZZ056, D.L.).
Authorship
WQ, LFR designed and performed research, analyzed data, and prepared the manuscript. ZMC, ZPT, XC, LYY, BL, LYH, YY and WF performed research. LD and FY designed research, analyzed data, and prepared the manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13(3):159-175
2. Wright HL, Moots RJ, Edwards SW. The multifactorial role of neutrophils in rheumatoid arthritis. Nat Rev Rheumatol. 2014;10(10):593-601
3. Nauseef WM. Myeloperoxidase in human neutrophil host defence. Cell Microbiol. 2014;16(8):1146-1155
4. Rodriguez-Espinosa O, Rojas-Espinosa O, Moreno-Altamirano MM, Lopez-Villegas EO, Sanchez-Garcia FJ. Metabolic requirements for neutrophil extracellular traps formation. Immunology. 2015;145(2):213-224
5. de Bruin AM, Libregts SF, Valkhof M, Boon L, Touw IP, Nolte MA. IFNgamma induces monopoiesis and inhibits neutrophil development during inflammation. Blood. 2012;119(6):1543-1554
6. Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 2006;6(3):173-182
7. Mollet L, Robinet P, Dubois M. et al. Opposing Mcl-1, the GALIG proapoptotic gene is upregulated as neutrophils die and underexpressed in Acute Myeloid Leukemia cells. Mol Immunol. 2013;56(1-2):123-128
8. Chen X, Xie H, Wood BL. et al. Relation of clinical response and minimal residual disease and their prognostic impact on outcome in acute myeloid leukemia. J Clin Oncol. 2015;33(11):1258-1264
9. Tsai S, Collins SJ. A dominant negative retinoic acid receptor blocks neutrophil differentiation at the promyelocyte stage. Proc Natl Acad Sci U S A. 1993;90(15):7153-7157
10. Hu XX, Zhong L, Zhang X, Gao YM, Liu BZ. NLS-RARalpha promotes proliferation and inhibits differentiation in HL-60 cells. Int J Med Sci. 2014;11(3):247-254
11. Li D, Yang H, Nan H. et al. Identification of key regulatory pathways of myeloid differentiation using an mESC-based karyotypically normal cell model. Blood. 2012;120(24):4712-4719
12. Allen IC, Wilson JE, Schneider M. et al. NLRP12 suppresses colon inflammation and tumorigenesis through the negative regulation of noncanonical NF-kappaB signaling. Immunity. 2012;36(5):742-754
13. Krauss JL, Zeng R, Hickman-Brecks CL, Wilson JE, Ting JP, Novack DV. NLRP12 provides a critical checkpoint for osteoclast differentiation. Proc Natl Acad Sci U S A. 2015;112(33):10455-10460
14. Lich JD, Williams KL, Moore CB. et al. Monarch-1 suppresses non-canonical NF-kappaB activation and p52-dependent chemokine expression in monocytes. J Immunol. 2007;178(3):1256-1260
15. Vaira S, Johnson T, Hirbe AC. et al. RelB is the NF-kappaB subunit downstream of NIK responsible for osteoclast differentiation. Proc Natl Acad Sci U S A. 2008;105(10):3897-3902
16. Hayden MS, Ghosh S. Regulation of NF-kappaB by TNF family cytokines. Semin Immunol. 2014;26(3):253-266
17. Zhao W, Bonem M, McWhite C, Silberg JJ, Segatori L. Sensitive detection of proteasomal activation using the Deg-On mammalian synthetic gene circuit. Nat Commun. 2014;5:3612
18. Li D, Hu J, Wang T. et al. Silymarin attenuates cigarette smoke extract-induced inflammation via simultaneous inhibition of autophagy and ERK/p38 MAPK pathway in human bronchial epithelial cells. Sci Rep. 2016;6:37751
19. Lupfer C, Kanneganti TD. The expanding role of NLRs in antiviral immunity. Immunol Rev. 2013;255(1):13-24
20. Strober W, Murray PJ, Kitani A, Watanabe T. Signalling pathways and molecular interactions of NOD1 and NOD2. Nat Rev Immunol. 2006;6(1):9-20
21. Futosi K, Fodor S, Mocsai A. Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int. Immunopharmacol. 2013;17(3):638-650
22. Avellino R, Delwel R. Expression and regulation of C/EBPalpha in normal myelopoiesis and in malignant transformation. Blood. 2017;129(15):2083-2091
23. Federzoni EA, Valk PJ, Torbett BE. et al. PU.1 is linking the glycolytic enzyme HK3 in neutrophil differentiation and survival of APL cells. Blood. 2012;119(21):4963-4970
24. Maun NA, Gaines P, Khanna-Gupta A. et al. G-CSF signaling can differentiate promyelocytes expressing a defective retinoic acid receptor: evidence for divergent pathways regulating neutrophil differentiation. Blood. 2004;103(5):1693-1701
25. Wang GG, Calvo KR, Pasillas MP, Sykes DB, Hacker H, Kamps MP. Quantitative production of macrophages or neutrophils ex vivo using conditional Hoxb8. Nat Methods. 2006;3(4):287-293
26. Lechtenberg BC, Mace PD, Riedl SJ. Structural mechanisms in NLR inflammasome signaling. Curr Opin Struct Biol. 2014;29:17-25
27. Thaiss CA, Elinav E. NF-kappaB Regulation by NLRs: T Cells Join the Club. Immunity. 2015;42(4):595-597
28. Vladimer GI, Marty-Roix R, Ghosh S, Weng D, Lien E. Inflammasomes and host defenses against bacterial infections. Curr Opin Microbiol. 2013;16(1):23-31
29. Zaki MH, Vogel P, Malireddi RK. et al. The NOD-like receptor NLRP12 attenuates colon inflammation and tumorigenesis. Cancer Cell. 2011;20(5):649-660
30. Zaki MH, Man SM, Vogel P, Lamkanfi M, Kanneganti TD. Salmonella exploits NLRP12-dependent innate immune signaling to suppress host defenses during infection. Proc Natl Acad Sci U S A. 2014;111(1):385-390
31. Zamoshnikova A, Groß CJ, Schuster S, Chen KW, Wilson A, Tacchini-Cottier F, Schroder K. NLRP12 is a neutrophil-specific, negative regulator of in vitro cell migration but does not modulate LPS- or infection-induced NF-κB or ERK signalling. Immunobiology. 2016;221(2):341-346
32. Lu T, Stark GR. NF-kappaB: Regulation by Methylation. Cancer Res. 2015;75(18):3692-3695
33. D'Ignazio L, Bandarra D, Rocha S. NF-kappaB and HIF crosstalk in immune responses. FEBS J. 2016;283(3):413-424
34. Lukens JR, Gurung P, Shaw PJ. et al. The NLRP12 Sensor Negatively Regulates Autoinflammatory Disease by Modulating Interleukin-4 Production in T Cells. Immunity. 2015;42(4):654-664
35. Chang JS, Santhanam R, Trotta R. et al. High levels of the BCR/ABL oncoprotein are required for the MAPK-hnRNP-E2 dependent suppression of C/EBPalpha-driven myeloid differentiation. Blood. 2007;110(3):994-1003
Author contact
Corresponding authors: Dong Li, Department of Oncology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China; E-mail: lidongedu.cn. and Yong Fang, Department of Burns and Plastic Surgery, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China; E-mail: fangyong1020com.

 Global reach, higher impact
Global reach, higher impact