Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(2):228-238. doi:10.7150/ijbs.38431 This issue Cite
Research Paper
Silencing of brain-expressed X-linked 2 (BEX2) promotes colorectal cancer metastasis through the Hedgehog signaling pathway
1. Department of Colorectal Surgery, The Second Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou, Zhejiang 310009, China.
2. Department of Medical Oncology, The Second Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou, Zhejiang 310009, China.
3. Cancer Institute (Key Laboratory of Cancer Prevention and Intervention, China National Ministry of Education, Key Laboratory of Molecular Biology in Medical Sciences, Zhejiang Province, China), The Second Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou, Zhejiang 310009, China.
*These authors contributed equally to this work and should be considered as first authors.
Abstract
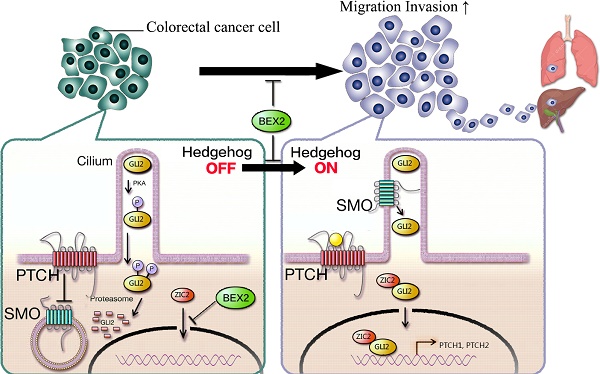
The incidence of colorectal cancer is increasing, and cancer metastasis is one of the major causes of poor outcomes. BEX2 has been reported to be involved in tumor development in several types of cancer, but its role in metastatic colorectal cancer remains largely undefined. Herein, we demonstrated that BEX2 knockout resulted in enhanced migratory and metastatic potential in colorectal cancer cells both in vitro and in vivo, and re-expression of BEX2 in knockout cells could reverse the enhanced migratory capacity. RNA-Seq results indicated that the hedgehog signaling pathway was activated after BEX2 knockout; moreover, the hedgehog signaling inhibitors, GANT61 and GDC-0449 could reverse the migratory enhancement of BEX2-/- colorectal cancer cells. We also demonstrated that the nuclear translocation of Zic2 after BEX2 silencing could activate the hedgehog signaling pathway, while Zic2 knockdown abrogated the migratory enhancement of BEX2-/- cells and inhibited the hedgehog signaling pathway. In summary, our findings suggest that BEX2 negatively modulates the hedgehog signaling pathway by retaining Zic2 in the cytoplasm in colorectal cancer cells, thereby inhibiting migration and metastasis of colorectal cancer cells.
Keywords: Colorectal cancer, BEX2, Zic2, Migration, Metastasis, Hedgehog signaling

 Global reach, higher impact
Global reach, higher impact