Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(7):1682-1692. doi:10.7150/ijbs.56251 This issue Cite
Review
Regulation of RNA N6-methyladenosine modification and its emerging roles in skeletal muscle development
1. Guangdong Provincial Key Laboratory of Animal Molecular Design and Precise Breeding, Key Laboratory of Animal Molecular Design and Precise Breeding of Guangdong Higher Education Institutes, School of Life Science and Engineering, Foshan University, Foshan 528231, Guangdong, China.
2. Shenzhen Branch, Guangdong Laboratory for Lingnan Modern Agriculture, Agricultural Genomics Institute at Shenzhen, Chinese Academy of Agricultural Sciences, Shenzhen, 518124, China.
3. State Key Laboratory of Animal Nutrition; Key Laboratory of Animal Genetics Breeding and Reproduction, Ministry of Agriculture and Rural Affairs, Institute of Animal Science, Chinese Academy of Agricultural Sciences, Beijing 100193, China.
Abstract
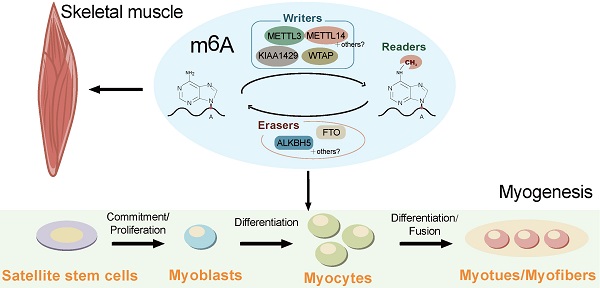
N6-methyladenosine (m6A) is one of the most widespread and highly conserved chemical modifications in cellular RNAs of eukaryotic genomes. Owing to the development of high-throughput m6A sequencing, the functions and mechanisms of m6A modification in development and diseases have been revealed. Recent studies have shown that RNA m6A methylation plays a critical role in skeletal muscle development, which regulates myoblast proliferation and differentiation, and muscle regeneration. Exploration of the functions of m6A modification and its regulators provides a deeper understanding of the regulatory mechanisms underlying skeletal muscle development. In the present review, we aim to summarize recent breakthroughs concerning the global landscape of m6A modification in mammals and examine the biological functions and mechanisms of enzymes regulating m6A RNA methylation. We describe the interplay between m6A and other epigenetic modifications and highlight the regulatory roles of m6A in development, especially that of skeletal muscle. m6A and its regulators are expected to be targets for the treatment of human muscle-related diseases and novel epigenetic markers for animal breeding in meat production.
Keywords: RNA N6-methyladenosine, development, skeletal muscle, myogenesis

 Global reach, higher impact
Global reach, higher impact