Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(8):1878-1894. doi:10.7150/ijbs.58657 This issue Cite
Research Paper
Protopanaxadiol improves endometriosis associated infertility and miscarriage in sex hormones receptors-dependent and independent manners
1. NHC Key Lab of Reproduction Regulation (Shanghai Institute of Planned Parenthood Research), Hospital of Obstetrics and Gynecology, Shanghai Medical School, Fudan University, Shanghai 200080, People's Republic of China.
2. Laboratory for Reproductive Immunology, Hospital of Obstetrics and Gynecology, Shanghai Medical School, Fudan University, Shanghai 200080, People's Republic of China.
3. Department of Gynecology, Hospital of Obstetrics and Gynecology, Shanghai Medical School, Fudan University, Shanghai 200011, People's Republic of China.
4. Assisted Reproductive Technology Unit, Department of Obstetrics and Gynecology, Faculty of Medicine, Chinese University of Hong Kong, Hong Kong, People's Republic of China.
5. Division of Obstetrics and Gynecology, KK Women's and Children's Hospital, 229899, Singapore.
6. National Research Centre for Carbohydrate Synthesis, Jiangxi Normal University, 330022 Jiangxi, Nanchang, China.
7. Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases, Hospital of Obstetrics and Gynecology, Shanghai Medical School, Fudan University, Shanghai, 200080, People's Republic of China.
Abstract
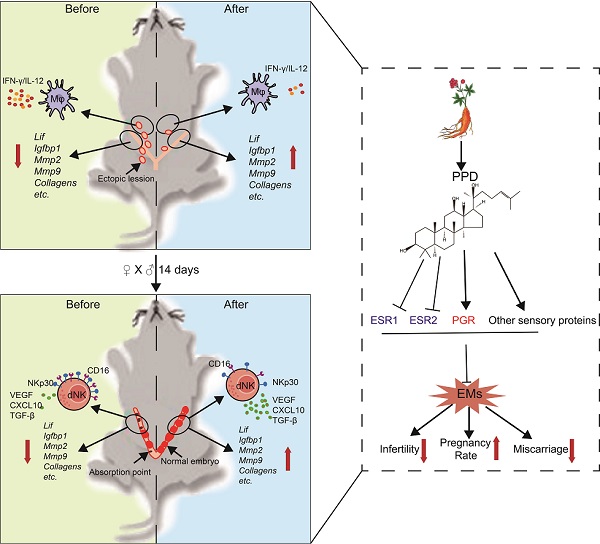
Background: Patients with endometriosis (EMs) have high risks of infertility and spontaneous abortion. How to remodel the fertility of patients with EMs has always been the hot spot and difficulty in the field of reproductive medicine. As an aglycone of ginsenosides, protopanaxadiol (PPD) possesses pleiotropic biological functions and has high medicinal values. We aimed to investigate the effect and potential mechanism of PPD in the treatment of EMs-associated infertility and spontaneous abortion.
Methods: The EMs mice models were constructed by allotransplantation. The pregnancy rates, embryo implantation numbers and embryo resorption rates of control and EMs were counted. RNA sequencing, qRT-PCR, enzyme linked immunosorbent assay (ELISA) and FCM analysis were performed to screen and confirm the expression of endometrial receptivity/decidualization-related molecules, inflammation cytokines and NK cell function-related molecules in vitro and/or in vivo. The SWISS Target Prediction, STRING and Cytoscape were carried out to predict the potential cellular sensory proteins, the protein-protein interaction (PPI) network between sensory proteins and fertility-related molecules, respectively. Micro-CT detection, liver and kidney function tests were used to evaluate the safety.
Results: Here, we observe that PPD significantly up-regulates endometrial receptivity-related molecules (e.g., Lif, Igfbp1, Mmps, collagens) and restricts pelvic inflammatory response (low levels of IL-12 and IFN-γ) of macrophage, and further remodel and improve the fertility of EMs mice. Additionally, PPD increases the expression of decidualization-related genes and Collagens, and promotes the proliferation, residence, immune tolerance and anagogic functions of decidual NK cells (low levels of CD16 and NKp30, high levels of Ki67, VEGF, TGF-β) in pregnant EMs mice, and further triggers decidualization, decidual NK cell-mediated maternal-fetal immune tolerance and angiogenesis, preventing pregnant EMs mice from miscarriage. Mechanically, these effects should be dependent on ESRs, PGR and other sensory proteins (e.g., AR). Compared with GnRHa (the clinic first-line drug for EMs), PPD does not lead to the decline of serum estrogen and bone loss.
Conclusion: These data suggest that PPD prevents EMs-associated infertility and miscarriage in sex hormones receptors-dependent and independent manners possibly, and provides a potential therapeutic strategy with high efficiency and low side effects to remodels the fertility of patients with EMs.
Keywords: endometriosis, protopanaxadiol, endometrial receptivity, decidualization, GnRHa

 Global reach, higher impact
Global reach, higher impact