10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(10):2487-2503. doi:10.7150/ijbs.60384 This issue Cite
Review
Towards Post-Meiotic Sperm Production: Genetic Insight into Human Infertility from Mouse Models
1. Division of Life Sciences and Medicine, The First Affiliated Hospital of USTC, University of Science and Technology of China, Anhui, China.
2. The 901th hospital of Joint logistics support Force of PLA, Anhui, China.
3. NHC Key Laboratory of Male Reproduction and Genetics, Family Planning Research Institute of Guangdong Province, China.
Abstract
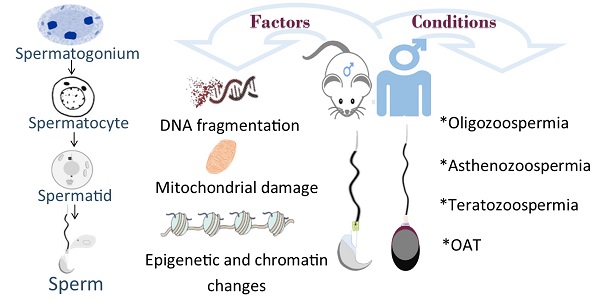
Declined quality and quantity of sperm is currently the major cause of patients suffering from infertility. Male germ cell development is spatiotemporally regulated throughout the whole developmental process. While it has been known that exogenous factors, such as environmental exposure, diet and lifestyle, et al, play causative roles in male infertility, recent progress has revealed abundant genetic mutations tightly associated with defective male germline development. In mammals, male germ cells undergo dramatic morphological change (i.e., nuclear condensation) and chromatin remodeling during post-meiotic haploid germline development, a process termed spermiogenesis; However, the molecular machinery players and functional mechanisms have yet to be identified. To date, accumulated evidence suggests that disruption in any step of haploid germline development is likely manifested as fertility issues with low sperm count, poor sperm motility, aberrant sperm morphology or combined. With the continually declined cost of next-generation sequencing and recent progress of CRISPR/Cas9 technology, growing studies have revealed a vast number of disease-causing genetic variants associated with spermiogenic defects in both mice and humans, along with mechanistic insights partially attained and validated through genetically engineered mouse models (GEMMs). In this review, we mainly summarize genes that are functional at post-meiotic stage. Identification and characterization of deleterious genetic variants should aid in our understanding of germline development, and thereby further improve the diagnosis and treatment of male infertility.
Keywords: Spermiogenesis, spermatogenesis, infertility, genetically engineered mouse model (GEMM), oligoasthenoteratozoospermia (OAT)

 Global reach, higher impact
Global reach, higher impact