Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(13):3369-3380. doi:10.7150/ijbs.61439 This issue Cite
Review
The role of RNA m5C modification in cancer metastasis
1. Hepatic Surgery Center, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430030, China.
2. Hubei Province for the Clinical Medicine Research Center of Hepatic Surgery, Wuhan, Hubei 430030, China.
3. Hubei key laboratory of Hepato-Pancreato-Biliary Diseases, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430030, China.
† Qiaofeng Zhang and Furong Liu contributed equally to this work.
Abstract
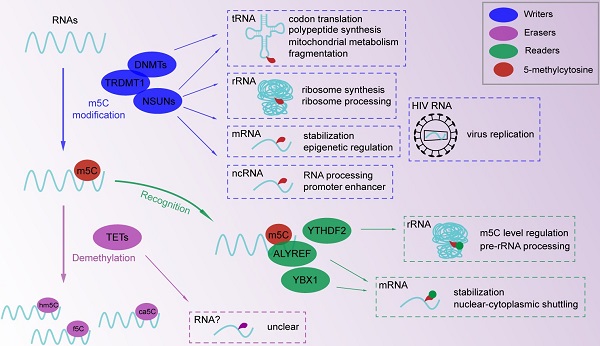
Epigenetic modification plays a crucial regulatory role in the biological processes of eukaryotic cells. The recent characterization of DNA and RNA methylation is still ongoing. Tumor metastasis has long been an unconquerable feature in the fight against cancer. As an inevitable component of the epigenetic regulatory network, 5-methylcytosine is associated with multifarious cellular processes and systemic diseases, including cell migration and cancer metastasis. Recently, gratifying progress has been achieved in determining the molecular interactions between m5C writers (DNMTs and NSUNs), demethylases (TETs), readers (YTHDF2, ALYREF and YBX1) and RNAs. However, the underlying mechanism of RNA m5C methylation in cell mobility and metastasis remains unclear. The functions of m5C writers and readers are believed to regulate gene expression at the post-transcription level and are involved in cellular metabolism and movement. In this review, we emphatically summarize the recent updates on m5C components and related regulatory networks. The content will be focused on writers and readers of the RNA m5C modification and potential mechanisms in diseases. We will discuss relevant upstream and downstream interacting molecules and their associations with cell migration and metastasis.
Keywords: Metastasis, cell migration, 5-methylcytosine, ribosome acid, regulatory network

 Global reach, higher impact
Global reach, higher impact