ISSN: 1449-2288
Int J Biol Sci 2022; 18(4):1508-1520. doi:10.7150/ijbs.68861 This issue Cite
Research Paper
Identification of Gαi3 as a promising target for osteosarcoma treatment
1. Department of Orthopedics, Clinical Research Center of Neurological Disease, The Second Affiliated Hospital of Soochow University, Suzhou, China.
2. Department of Orthopedics, The Affiliated Hospital of Yangzhou University, Yangzhou University, Yangzhou, China.
3. Department of Orthopedics, Affiliated Jiangyin Hospital of Xuzhou Medical University, Jiangyin, China.
4. Department of Orthopedics, The Affiliated Suqian Hospital of Xuzhou Medical University, Suqian, China.
5. Department of Radiotherapy and Oncology, Kunshan First People's Hospital Affiliated to Jiangsu University, Kunshan, China.
6. Department of Orthopedics, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China.
7. Department of Orthopedics, Children's Hospital of Soochow University, Suzhou, China.
8. Jiangsu Key Laboratory of Neuropsychiatric Diseases and Institute of Neuroscience, Soochow University, Suzhou, China.
9. North District, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou Municipal Hospital, Suzhou, China.
# Co-first authors.
Abstract
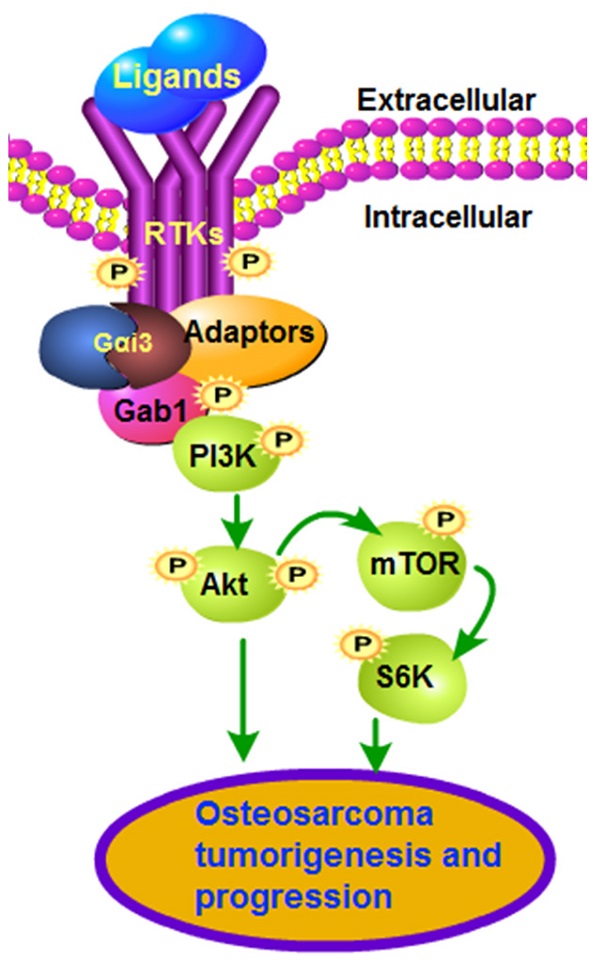
Sustained activation of multiple receptor tyrosine kinases (RTKs) simultaneously is vital for tumorigenesis and progression of osteosarcoma (OS). Gαi proteins recruitment to various RTKs mediates downstream oncogenic signaling activation. The expression, functions and underlying mechanisms of Gαi3 in human OS were examined. Expression of Gαi3 is robustly elevated in human OS tissues and is correlated with a poor overall survival. In patient-derived primary OS cells and immortalized lines (MG63 and U2OS), Gαi3 depletion, by shRNA and CRISPR/Cas9 strategies, robustly suppressed cell viability, proliferation and migration, while provoking G1-S arrest and apoptosis activation. Conversely, Gαi3 overexpressing ectopically can accelerate proliferation and migration of OS cells. In OS cells, Gαi3 immunoprecipitated with VEGFR2, FGFR, PGDFR and EGFR, mediating downstream cascade transduction. Akt-mTOR activation in primary OS cells was potently inhibited by Gαi3 shRNA, knockout or dominant negative mutation, but augmented after Gαi3 overexpression. In vivo studies showed that Gαi3 shRNA AAV (adeno-associated viruses) intratumoral injection largely inhibited the growth of subcutaneous xenografts of primary OS cells. Moreover, the growth of the Gαi3-knockout primary OS xenografts was much slower than that of OS xenografts with empty vector. In Gαi3-depleted OS xenografts tissues, Gαi3 downregulation and Akt-mTOR inactivation were detected. Taken together, overexpressed Gαi3 mediates RTK-Akt signaling to drive OS progression.
Keywords: Osteosarcoma, Gαi3, multiple receptor tyrosine kinase, Akt-mTOR, Signaling

 Global reach, higher impact
Global reach, higher impact