Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2023; 19(3):760-771. doi:10.7150/ijbs.77495 This issue Cite
Research Paper
The ADAM9/WISP-1 axis cooperates with osteoblasts to stimulate primary prostate tumor growth and metastasis
1. Translational Medicine Center, Shin Kong Wu Ho-Su Memorial Hospital, Taipei, Taiwan.
2. Graduate Institute of Biomedical Sciences, China Medical University, Taichung, Taiwan.
3. Department of Life Science, National Taiwan Normal University, Taipei, Taiwan.
4. Department of Medical Research, China Medical University Hospital, China Medical University, Taichung, Taiwan.
5. Department of Medical Education and Research, China Medical University Beigang Hospital, Yunlin, Taiwan.
6. School of Medicine, Fu-Jen Catholic University, New Taipei City, Taiwan.
7. Department of Urology, Fu-Jen Catholic University Hospital, New Taipei City, Taiwan.
8. School of Medicine, China Medical University, Taichung, Taiwan.
9. Department of Urology, China Medical University Hospital, Taichung, Taiwan.
10. Department of Urology, China Medical University Beigang Hospital, Beigang, Yunlin, Taiwan.
11. Graduate Institute of Clinical Medicine, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan.
12. International Ph.D. Program for Translational Science, College of Medical Science and Technology, Taipei Medical University, Taipei, Taiwan.
13. The Ph.D. Program for Translational Medicine, College of Medical Science and Technology, Taipei Medical University, Taipei, Taiwan.
14. TMU Research Center of Cancer Translational Medicine, Taipei Medical University, Taipei, Taiwan.
15. Office of Human Research, Taipei Medical University, Taipei, Taiwan.
16. TMU-Research Center of Urology and Kidney, Taipei Medical University, Taipei, Taiwan.
17. Department of Urology, Buddhist Tzu Chi General Hospital Taichung Branch, Taichung, Taiwan.
18. School of Post-Baccalaureate Chinese Medicine, Tzu Chi University, Hualien, Taiwan.
19. Chinese Medicine Research Center, China Medical University, Taichung, Taiwan.
20. Department of Biotechnology, Asia University, Taichung, Taiwan.
Abstract
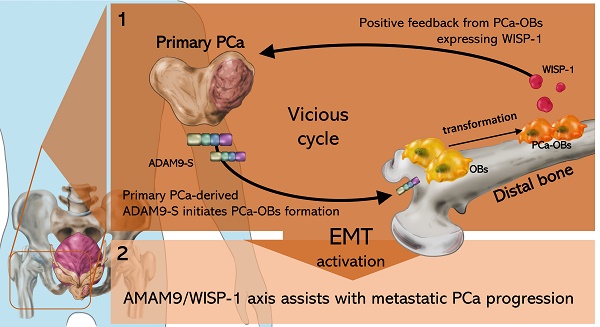
Background: Metastatic prostate cancer (PCa) predicts a poor prognosis and lower likelihood of survival. Osteoblasts (OBs) are known to be responsible for the synthesis and mineralization of bone, although it is unclear as to whether PCa in the prostate gland cooperates with OBs in bone to promote PCa malignant transformation. We aimed to elucidate how primary PCa cells cooperate with distal OBs and contribute to the vicious cycle that leads to metastatic PCa.
Methods: N-cadherin, E-cadherin, and Twist protein expression were measured by Western blot. Twist translocation into the nucleus was detected by the immunofluorescence (IF) assay. Enzyme-linked immunosorbent assay (ELISA) detected protein levels in human serum samples. Levels of candidate protein expression were examined by the human cytokine array. Prostate tumor growth and metastasis were analyzed by orthotopic and metastatic prostate cancer models, respectively. Immunohistochemistry (IHC) staining was used to observe ADAM metallopeptidase domain 9 (ADAM9) and WNT1 inducible signaling pathway protein 1 (WISP-1) expression in tissue.
Results: Our in vitro and in vivo analyses have now discovered that primary PCa expressing ADAM9 protein enables the transformation of OBs into PCa-associated osteoblasts (PCa-OBs), inducing WISP-1 secretion from PCa-OBs in the bone microenvironment. The upregulation of WISP-1 in bone provided feedback to primary PCa and promoted PCa cell aggressiveness via epithelial-mesenchymal transition (EMT) activity. Elevated levels of WISP-1 expression were detected in the serum of patients with PCa. ADAM9 levels were overexpressed in tumor tissue from PCa patients; ADAM9 blockade interrupted OB-induced release of WISP-1 and also suppressed primary tumor growth and distal metastasis in orthotopic PCa mouse models.
Conclusion: Our study suggests that the ADAM9/WISP-1 axis assists with metastatic PCa progression. Thus, targeting the ADAM9/WISP-1 axis may help to prevent the malignant phenotypes of PCa cells.
Keywords: Prostate cancer, Osteoblast, WISP-1, ADAM9, Metastasis

 Global reach, higher impact
Global reach, higher impact